- - Google Chrome
Intended for healthcare professionals
- My email alerts
- BMA member login
- Username * Password * Forgot your log in details? Need to activate BMA Member Log In Log in via OpenAthens Log in via your institution


Search form
- Advanced search
- Search responses
- Search blogs
- News & Views
- Decision aids that...
Decision aids that really promote shared decision making: the pace quickens
- Related content
- Peer review
- Thomas Agoritsas , research fellow 1 2 ,
- Anja Fog Heen , doctoral candidate 3 4 ,
- Linn Brandt , doctoral candidate 3 4 ,
- Pablo Alonso-Coello , associate researcher 1 5 ,
- Annette Kristiansen , doctoral candidate 3 4 ,
- Elie A Akl , associate professor 1 6 ,
- Ignacio Neumann , assistant professor 1 7 ,
- Kari AO Tikkinen , adjunct professor 1 8 ,
- Trudy van der Weijden , professor 9 ,
- Glyn Elwyn , professor 10 ,
- Victor M Montori , professor 11 ,
- Gordon H Guyatt , distinguished professor 1 ,
- Per Olav Vandvik , associate professor 3 4
- 1 Department of Clinical Epidemiology and Biostatistics, McMaster University, Faculty of Health Sciences, Hamilton, Ontario, Canada
- 2 Division of General Internal Medicine, Division of Clinical Epidemiology, University Hospitals of Geneva, Switzerland
- 3 Department of Medicine, Innlandet Hospital Trust, Gjøvik, Norway
- 4 Institute for Health and Society, Faculty of Medicine, University of Oslo, Oslo, Norway
- 5 Iberoamerican Cochrane Centre, Biomedical Research Institute Sant Pau—CIBER, Epidemiología y Salud Pública, Barcelona, Spain
- 6 Department of Internal Medicine, American University of Beirut, Lebanon
- 7 Department of Internal Medicine, School of Medicine, Pontificia Universidad Católica de Chile, Santiago, Chile
- 8 Departments of Urology and Public Health, Helsinki University Central Hospital and University of Helsinki, Helsinki, Finland
- 9 Department Family Medicine, School for Public Health and Primary Care, Maastricht University, Maastricht, Netherlands
- 10 Dartmouth Center for Health Care Delivery Science, Dartmouth Institute for Health Policy and Clinical Practice, Hanover, USA
- 11 Knowledge and Evaluation Research Unit, Mayo Clinic, Rochester, USA
- Correspondence to: T Agoritsas, thomas.agoritsas{at}gmail.com
Decision aids can help shared decision making, but most have been hard to produce, onerous to update, and are not being used widely. Thomas Agoritsas and colleagues explore why and describe a new electronic model that holds promise of being more useful for clinicians and patients to use together at the point of care
Many, perhaps most, important decisions in medicine are not clear cut. 1 2 Patients and clinicians need to discuss the options using the best available evidence and make informed joint decisions that take account of patients’ context, values, and preferences. 3 4 But implementing shared decision making is not easy. Doctors need the skills and tools to do it and to build trust; patients need information and support. Patients also need to have a greater role in developing strategies to improve the process. 5 6
Access to best evidence is another key ingredient. Until now the production and dissemination of clinical practice guidelines and summaries of evidence has largely been tailored to meet the educational needs of clinicians. They are seldom provided in a format that supports shared decision making. 7 Patients meanwhile, struggle to find reliable and accessible summaries of evidence, although plain language summaries and patient versions of guidelines are being developed. 8
In this article we highlight the limitations of current decision aids and discuss how the generic production of electronic decision aids designed for use in the clinical encounter, linked directly to trustworthy summaries of evidence from systematic reviews and guidelines, may help in the long march to realising effective shared decision making.
Challenge of shared decision making
Shared decision making depends on a good conversation 9 in which clinicians share information about the benefits, harms, and burden of alternative diagnostic and therapeutic options and patients explain what matters to them and their views on the choices they face. 4 10 It should follow the principles of patient centred care, promote informed choice, and result in care that patients value. 1 3 11 Many clinicians think they practice shared decision making, but evidence suggest a perception-reality gap 3 because of misconceptions about the nature of shared decision making, the skills it requires, the time it takes, and the degree to which patients, families, and carers wish to share in decision making. 12 13 14
Each clinical encounter is influenced by many factors. These include patients’ circumstances and medical needs as well as their beliefs, stemming from what they have read, personal experience, advice from family and friends, and the media. It is therefore important to provide patients with accurate, up to date evidence on the benefits and harms of alternative management strategies and their likely effect on outcomes that matter to them, although evidence may not always reflect the complexity and multimorbidity of individual patients and patients may choose to ignore the evidence . Good shared decision making requires clinicians to have access to detailed knowledge and ideally summaries of the latest evidence and the means to share it in a way that supports thoughtful deliberation, something that cannot be done on the fly.
Limitations of traditional decision aids
For the past two decades enthusiasts have advocated decision aids to facilitate shared decision making, and over 500 have been developed. 15 16 A systematic review of 115 randomised trials showed that their use was associated with a 13% absolute increase in patients’ knowledge scores and an 82% relative increase in accurate expectations of possible benefits and harms. Effects on clinical outcomes, adherence to treatment, and use of services have not, however, been consistent. 15 17
Most decision aids have been designed for patients to use independently outside the consultation, either in the waiting room or at home. 10 Although these decision aids promote understanding of the issues, they cannot guarantee that decisions in the consultation are shared, 3 18 and there is insufficient evidence to determine how their use influences the consultation. 18 Another problem is that use of decision aids in routine care is low, 13 mainly because of poor design and lack of ready access to them. Furthermore clinicians may find the format impractical to use in consultations and may be as unfamiliar as their patients with risk estimates and the inherent uncertainty associated with probabilities. 19
Traditional decision aids are often not based on current evidence or rapidly outdated, at least in part because of limitations in funding after tool development—and may thus do more harm than good. 20 A rigorous systematic review is needed for each important outcome, and such reviews are often unavailable. A recent assessment found that although around two thirds of decision aids are based on systematic reviews or guidelines, many of these sources are of questionable quality, and only 5% of aids included an “expiry date” or a stated policy about updating. 20
Ensuring the quality and timeliness of decision aids is a daunting challenge. The work required to summarise evidence for a trustworthy decision aid is similar to that for producing a systematic review or a guideline, suggesting the potential for synergy between the worlds of evidence based practice and shared decision making. 20 21 22
Harnessing the potential of recent developments
New decision aids.
Some newer decision aids have been designed to facilitate collaborative deliberation in the course of the clinical encounter. 3 10 Montori and colleagues pioneered a user centred approach to producing decision aids through iterative observations of discussions between doctors and patients. 9 23 Their approach resulted in succinct, easy to use tools that provide graphic displays of the benefits and harms of different options organised around concerns that are important to patients ( http://shareddecisions.mayoclinic.org ). In contrast to traditional aids, which patients use independently, they are not designed to be comprehensive and do not include explicit exercises to help patients clarify their values (such as the relative values of avoiding a stroke versus a gastrointestinal bleed) 24 Instead they rely on the unique conversations that take place between patients and clinicians, with clinicians providing just in time, tailored explanations and information. 10 Direct observations in randomised trials have shown that these short tools (so far available for diabetes, statins, and antidepressants) promote dialogue and increase joint deliberation. 25 They also shift the “body language” as patients and clinicians sit together to review the data. 23 26
Other short point of care decision aids include Option Grids ( www.optiongrid.co.uk ). 27 28 These are one page summaries that provide answers to patients’ frequently asked questions, covering clinical outcomes and practical concerns faced in daily life. Their value in routine care is being evaluated. 27
Developments in appraisal and presentation of best evidence
The GRADE approach (Grading of Recommendations Assessment, Development and Evaluation) provides systematic, transparent, and explicit guidance for processing evidence from the medical literature, and has been widely adopted. 7 29 30 Use of the GRADE approach results in standardised and succinct evidence profiles or summary of findings tables, which specify the absolute effects of an intervention on outcomes important to patients rather than surrogate outcomes and provide a rating of the certainty in these estimates (high, moderate, low, or very low). 30 The recent international patient decision aids standards have emphasised the potential of GRADE for the production of decision aids 20 , and it has been adopted by over 80 organisations ( www.gradeworkinggroup.org ).
Furthermore, clinical practice guidelines using GRADE now issue weak recommendations (in contrast to strong) when there is a close balance between desirable and undesirable outcomes among alternatives, low certainty in estimates of effect, or when there is large variability in patients’ values and preferences. Weak recommendations, which dominate in recent high quality guidelines, 2 thus identify decisions where shared decision making is particularly important. 20 22
Use of new technologies
The not-for-profit MAGIC project (Making GRADE the Irresistible Choice www.magicproject.org ) has developed an online “app” with potential to produce electronic decision aids for use in the clinical encounter. 7 This MAGICapp ( www.magicapp.org ) allows authors of guidelines or systematic reviewers to write evidence summaries into a structured database and appraise them using GRADE criteria. The content can then be published on a web platform and presented in interactive formats on tablets, web portals, or electronic medical record systems. 31
In the SHARE-IT project, we use this authoring and publication platform for the generic and semi-automated production of a large number of decision aids. 7 The aids can be used with the corresponding systematic review or clinical practice guidelines and the format modified and tailored to specific contexts—for example, published in different languages or adapted to national guidelines. 32 33 The electronic format facilitates continuous updating because the data in the decision aids will change automatically each time the underlying review is modified. 7
Figure 1 ⇓ summarises the methods of the SHARE-IT project. In collaboration with DECIDE ( www.decide-collaboration.eu ), 34 we gathered an international team of experts in evidence based medicine and shared decision making, clinicians, guideline developers, and designers, and developed an initial framework and electronic prototype for the translation of GRADE summaries into decision aids. We then applied an iterative and user centred design, directly involving patients and clinicians facing real decisions. We built 10 decision aids on antithrombotic drugs and modified the generic prototype in light of observations of their use in practice and individual feedback from patients and clinicians.
Fig 1 Outline of the methods and user-centred approach in the SHARE-IT project. Objective A=to develop a framework for the generic translation of GRADE evidence summaries into decision aids; Objective B=to design a set of interactive presentation formats for use in the clinical encounter; Objective C=to test the feasibility of an automated production of these decision aids from electronically published evidence summaries. Subsequent phases of the project involve the generic production of decision aids from real practice guidelines and their evaluation in randomised trials and cohort studies
- Download figure
- Open in new tab
- Download powerpoint
The video illustrates how the prototype uses interactive formats to present evidence summaries at varying levels of detail. The prototype shows that the approach is feasible, and preliminary experience suggests it is appreciated by both patients and clinicians (box). Across 16 clinical encounters, patients consistently reported high levels of satisfaction with the prototype in understanding risks and benefits and in enhancing their confidence in decisions (mean scores of 88.7 and 90.9 respectively (maximum 100) as assessed by COMRADE. 35
Reaction to the decision aid
A haematologist expressed surprise that one decision aid regarding long term anticoagulation treatment for patients with unprovoked venous thromboembolism begins by inviting patients to choose which outcome to discuss first. She usually started by discussing the risk of recurrence, then bleeding before inviting patients’ questions, omitting mortality.
After we clarified she could use the tool as she wanted, she began with the six month follow-up of a 47 year old man taking rivaroxaban for an unprovoked pulmonary embolism. She explained that, although the treatment was indicated after the acute event, the decision to continue rivaroxaban depended on his preferences. She accessed the decision aid and moved to sit next to the patient. Revising her prior plan to use her accustomed order, she used the trigger sentence offered: “What aspect of your medication would you like to discuss first?” The patient chose “practical consequences.” In the conversation that followed, they further discussed risk of bleeding, recurrence, and associated mortality. The patient decided to discontinue rivaroxaban.
After the encounter, the clinician pointed out that the patient focused on practical consequences first, and she reflected on how the tool resulted in positive changes to her usual communication strategy. The patient reported that the decision aid made it easier to “digest the information and get the bigger picture.” He explained he was first interested by “day-to-day stuff” before exploring “more intimidating” but important issues.
No decision aid is sufficient to guarantee that clinical decision making is shared. Undergraduate, postgraduate, and continuing education programmes must teach health professionals about the importance of creating and fostering a culture of shared decision making and the skills needed to communicate evidence, and its limitations, in a way people can understand. Furthermore, the challenge of producing evidence summaries that deal optimally with complexity, multimorbidity, and potentially limited applicability to the patient remains. 36
We are, however, now in a position to construct, test, and refine electronic evidence summaries for use in the clinical encounter for a wide variety of patient groups and clinical settings. Our prototype, built in the MAGICapp, demonstrates the feasibility of semiautomated production of decision aids from a large number of electronically published evidence summaries. We also plan to implement these formats in another similar platform, the GRADEpro Guideline Development Tool ( www.guidelinedevelopment.org ). We invite patient organisations, research groups, guideline developers, patients, and clinicians to partner with us ( www.magicproject.org ) and help us advance the science and art of truly shared and well informed decision making.
Cite this as: BMJ 2015;350:g7624
We thank Frankie Achille (interaction designer), Rob Fracisco (designer), and Deno Vichas and Chris Degiere (programmers) for their contributions in development of the online authoring and publication platform prototype ( www.magicproject.org ). TA was financially supported by a fellowship for prospective researchers grant No P3SMP3-155290/1 from the Swiss National Science Foundation, as well as by a fellowship grant from the University Hospitals of Geneva and from Eugenio Litta—Fondation Genevoise de Bienfaisance Valeria Rossi di Montelera. PA-C is funded by a Miguel Servet research contract from the Instituto de Salud Carlos III (CP09/00137). KAOT is funded by the Academy of Finland (#276046), Jane and Aatos Erkko Foundation, and Sigrid Jusélius Foundation. The Innlandet Hospital Trust, South-Eastern Norway Regional Health Authority and Innovation Norway have provided research grants for the MAGIC program ( www.magicproject.org ). This project has received funding from the European Union’s Seventh Framework Programme for research, technological development and dissemination under grant agreement No 258583. ( www.decide-collaboration.eu )
Contributors and sources: The SHARE-IT project was conceived and is mainly funded by the MAGIC program, in close collaboration with the DECIDE project and GRADE working group, to which most contributors are affiliated. We also received numerous feedbacks from stakeholders at international meetings. TA led and coordinated the project, supervised by GHG and POV. TA, AFH, LB, and POV developed and implemented the prototype, and all contributors provided feedback at different stages. TA, AFH, and POV performed user-testing in clinical encounters. TA drafted the manuscript and all authors critically revised the manuscript. TA is guarantor.
Competing interests: All authors have read and understood BMJ policy on declaration of interests and declare the following interests: TA, AFH, LB, AK, PAC, EAA, IN, KAOT, VMM, GHG, POV are members of the GRADE working group ( www.gradeworkinggroup.org ), as well as coinvestigators in the DECIDE project ( www.decide-collaboration.eu ). TA, AFH, LB, AK, GHG, POV are members of the MAGIC research and innovation program. GE leads the Option Grid collaborative. VMM designs and tests shared decision making tools at the KER UNIT in Mayo Clinic. These tools are then made available for free with no income generated for him, his unit, or his institution.
Provenance and peer review: Not commissioned; externally peer reviewed.
- ↵ Barry MJ, Edgman-Levitan S. Shared decision making--pinnacle of patient-centered care. N Engl J Med 2012 ; 366 : 780 -1. OpenUrl CrossRef PubMed Web of Science
- ↵ Djulbegovic B, Guyatt GH. Evidence-based practice is not synonymous with delivery of uniform health care. JAMA 2014 ; 312 : 1293 -4. OpenUrl CrossRef PubMed Web of Science
- ↵ Stiggelbout AM, Van der Weijden T, De Wit MP, Frosch D, Legare F, Montori VM, et al. Shared decision making: really putting patients at the centre of healthcare. BMJ 2012 ;344:e256.
- ↵ Elwyn G, Lloyd A, May C, van der Weijden T, Stiggelbout A, Edwards A, et al. Collaborative deliberation: a model for patient care. Patient Educ Counsel 2014 ; 97 : 158 -64. OpenUrl CrossRef PubMed
- ↵ Legare F, Ratte S, Stacey D, Kryworuchko J, Gravel K, Graham ID, et al. Interventions for improving the adoption of shared decision making by healthcare professionals. Cochrane Database Syst Rev 2010 ; 5 : CD006732 . OpenUrl PubMed
- ↵ Greenhalgh T, Howick J, Maskrey N. Evidence based medicine: a movement in crisis? BMJ 2014 ; 348 : g3725 . OpenUrl FREE Full Text
- ↵ Vandvik PO, Brandt L, Alonso-Coello P, Treweek S, Akl EA, Kristiansen A, et al. Creating clinical practice guidelines we can trust, use, and share: a new era is imminent. Chest 2013 ; 144 : 381 -9. OpenUrl CrossRef PubMed Web of Science
- ↵ Boivin A, Currie K, Fervers B, Gracia J, James M, Marshall C, et al. Patient and public involvement in clinical guidelines: international experiences and future perspectives. Qual Safe Health Care 2010 ; 19 : e22 . OpenUrl Abstract / FREE Full Text
- ↵ Montori VM, Breslin M, Maleska M, Weymiller AJ. Creating a conversation: insights from the development of a decision aid. PLoS Med 2007 ; 4 : e233 . OpenUrl CrossRef PubMed
- ↵ Elwyn G, Frosch D, Volandes AE, Edwards A, Montori VM. Investing in deliberation: a definition and classification of decision support interventions for people facing difficult health decisions. Med Decis Making 2010 ; 30 : 701 -11. OpenUrl Abstract / FREE Full Text
- ↵ Dwamena F, Holmes-Rovner M, Gaulden CM, Jorgenson S, Sadigh G, Sikorskii A, et al. Interventions for providers to promote a patient-centred approach in clinical consultations. Cochrane Database Syst Rev 2012 ; 12 : CD003267 . OpenUrl PubMed
- ↵ Hoffmann TC, Legare F, Simmons MB, McNamara K, McCaffery K, Trevena LJ, et al. Shared decision making: what do clinicians need to know and why should they bother? Med J Aust 2014 ; 201 : 35 -9. OpenUrl CrossRef PubMed Web of Science
- ↵ Elwyn G, Legare F, van der Weijden T, Edwards A, May C. Arduous implementation: does the Normalisation Process Model explain why it’s so difficult to embed decision support technologies for patients in routine clinical practice. Implement Sci 2008 ; 3 : 57 . OpenUrl CrossRef PubMed
- ↵ Cox K, Britten N, Hooper R, White P. Patients’ involvement in decisions about medicines: GPs’ perceptions of their preferences. Br J Gen Pract 2007 ; 57 : 777 -84. OpenUrl Abstract / FREE Full Text
- ↵ Stacey D, Legare F, Col NF, Bennett CL, Barry MJ, Eden KB, et al. Decision aids for people facing health treatment or screening decisions. Cochrane Database Syst Rev 2014 ; 1 : CD001431 . OpenUrl PubMed
- ↵ Patient Decision Aids Inventory. Ottawa Hospital Research Institute and University of Ottawa. http://decisionaid.ohri.ca/index.html .
- ↵ Walsh T, Barr PJ, Thompson R, Ozanne E, O’Neill C, Elwyn G. Undetermined impact of patient decision support interventions on healthcare costs and savings: systematic review. BMJ 2014 ; 348 : g188 . OpenUrl Abstract / FREE Full Text
- ↵ Hargraves I, Montori VM. Decision aids, empowerment, and shared decision making. BMJ 2014 ; 349 : g5811 . OpenUrl FREE Full Text
- ↵ Perneger TV, Agoritsas T. Doctors and patients’ susceptibility to framing bias: a randomized trial. J Gen Intern Med 2011 ; 26 : 1411 -7. OpenUrl CrossRef PubMed
- ↵ Montori VM, LeBlanc A, Buchholz A, Stilwell DL, Tsapas A. Basing information on comprehensive, critically appraised, and up-to-date syntheses of the scientific evidence: a quality dimension of the international patient decision aid standards. BMC Med Inform Decis Making 2013 ;13 Suppl 2:S5.
- ↵ Hoffmann TC, Montori VM, Del Mar C. The connection between evidence-based medicine and shared decision making. JAMA 2014 ; 312 : 1295 -6. OpenUrl CrossRef PubMed Web of Science
- ↵ Van der Weijden T, Boivin A, Burgers J, Schunemann HJ, Elwyn G. Clinical practice guidelines and patient decision aids. An inevitable relationship. J Clin Epidemiol 2012 ; 65 : 584 -9. OpenUrl CrossRef PubMed
- ↵ Breslin M, Mullan RJ, Montori VM. The design of a decision aid about diabetes medications for use during the consultation with patients with type 2 diabetes. Patient Educ Counsel 2008 ; 73 : 465 -72. OpenUrl CrossRef PubMed Web of Science
- ↵ Llewellyn-Thomas HA, Crump RT. Decision support for patients: values clarification and preference elicitation. Med Care Res Rev 2013 ; 70 (1 suppl): 50 -79S. OpenUrl CrossRef
- ↵ Coylewright M, Branda M, Inselman JW, Shah N, Hess E, LeBlanc A, et al. Impact of sociodemographic patient characteristics on the efficacy of decision aids: a patient-level meta-analysis of 7 randomized trials. Circ Cardiovasc Qual Outcomes 2014 ; 7 : 360 -7. OpenUrl Abstract / FREE Full Text
- ↵ Wyatt KD, Branda ME, Anderson RT, Pencille LJ, Montori VM, Hess EP, et al. Peering into the black box: a meta-analysis of how clinicians use decision aids during clinical encounters. Implement Sci 2014 ; 9 : 26 . OpenUrl CrossRef PubMed
- ↵ Lloyd A, Joseph-Williams N, Edwards A, Rix A, Elwyn G. Patchy ‘coherence’: using normalization process theory to evaluate a multi-faceted shared decision making implementation program (MAGIC). Implement Sci 2013;8:102.
- ↵ Giguere A, Labrecque M, Haynes R, Grad R, Pluye P, Legare F, et al. Evidence summaries (decision boxes) to prepare clinicians for shared decision-making with patients: a mixed methods implementation study. Implement Sci 2014 ; 9 : 144 . OpenUrl CrossRef PubMed
- ↵ Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, Alonso-Coello P, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ 2008 ; 336 : 924 -6. OpenUrl FREE Full Text
- ↵ Guyatt G, Oxman AD, Akl EA, Kunz R, Vist G, Brozek J, et al. GRADE guidelines: 1. Introduction-GRADE evidence profiles and summary of findings tables. J Clin Epidemiol 2011 ; 64 : 383 -94. OpenUrl CrossRef PubMed
- ↵ Kristiansen A, Brandt L, Alonso-Coello P, Agoritsas T, Akl EA, Conboy T, et al. Development of a novel multilayered presentation format for clinical practice guidelines. Chest 2014 Oct 9 [Epub ahead of print].
- ↵ Kristiansen A, Brandt L, Agoritsas T, Akl EA, Berge E, Bondi J, et al. Adaptation of trustworthy guidelines developed using the GRADE methodology: a novel 5-step process. Chest 2014 ; 146 : 727 -34. OpenUrl CrossRef PubMed Web of Science
- ↵ Kristiansen A, Brandt L, Agoritsas T, Akl EA, Berge E, Jacobsen AF, et al. Applying new strategies for the national adaptation, updating and dissemination of trustworthy guidelines: results from the Norwegian adaptation of the American College of Chest Physicians evidence-based guidelines on antithrombotic therapy and the prevention of thrombosis, 9th wdition. Chest 2014 ; 146 : 735 -61. OpenUrl CrossRef PubMed Web of Science
- ↵ Treweek S, Oxman AD, Alderson P, Bossuyt PM, Brandt L, Brozek J, et al. Developing and evaluating communication strategies to support informed decisions and practice based on evidence (DECIDE): protocol and preliminary results. Implement Sci 2013;8:6.
- ↵ Edwards A, Elwyn G, Hood K, Robling M, Atwell C, Holmes-Rovner M, et al. The development of COMRADE—a patient-based outcome measure to evaluate the effectiveness of risk communication and treatment decision making in consultations. Patient Educ Counsel 2003 ; 50 : 311 -22. OpenUrl CrossRef PubMed Web of Science
- ↵ Uhlig K, Leff B, Kent D, Dy S, Brunnhuber K, Burgers JS, et al. A framework for crafting clinical practice guidelines that are relevant to the care and management of people with multimorbidity. J Gen Intern Med 2014 ; 29 : 670 -9. OpenUrl CrossRef PubMed
University of Washington
Health Systems and Population Health
School of public health.
- The Role of Patient Decision Aids: Supporting High-Quality Patient-Centered Healthcare
- News & Events
Healthcare decisions are often complex, and, in many cases, the best choice depends on patient preferences and how individuals value the risks and benefits of different options. In such cases, it is recommended that patients and clinicians engage in shared decision-making, a collaborative process that considers the best available clinical evidence and the patients’ informed preferences. Patient decision aids are evidence-based tools to support shared decision-making. They include pamphlets, videos, or web-based resources that describe the decision, the options, and help people think about what features are most important to them. These evidence-based tools are specially designed to support individuals in making informed choices aligned with their values and preferences.
Dr. Logan Trenaman, an Assistant Professor in the Department of Health Systems and Population Health, is the co-author of the Cochrane Systematic Review of patient decision aids , which was updated and released in early 2024. According to Dr. Trenaman, the updated findings re-affirm what has been known for some time:
“Compared to usual care, patient decision aids increase patients’ knowledge, their participation in decision-making, and help them make decisions that are aligned with their preferences. Further, there is no evidence of negative effects, like increased decisional regret, associated with their use.” – Dr Logan Trenaman
The Cochrane Review of Patient Decision Aids:
This Cochrane review, initiated in 2003, plays a pivotal role in summarizing the existing evidence on the impact of these tools. It is the most referenced review in the Cochrane collection (over 8400 citations). In their 2024 update, Dr. Logan Trenaman and the Cochrane team identified 104 new randomized controlled trials of patient decision aids. This brings the total number of trials in the review to 209, spanning 71 different health care decisions (and including nearly 110,000 patients – enough to fill Husky Stadium one and half times).
Cochrane Reviews provide a comprehensive and systematic analysis of a specific healthcare topic and are conducted by the Cochrane Collaboration—an international network of researchers, healthcare professionals, patients, and policymakers. These reviews follow a rigorous methodology, and synthesize data from randomized controlled trials and other high-quality studies to provide a reliable overview of the available evidence. Cochrane Reviews can be valuable resources for healthcare professionals, policymakers, and patients, offering evidence-based insights that guide informed decision-making and contribute to advancements in medical knowledge and practice.
Implications and Future Directions

The implications of the findings for care delivery are profound. “All health systems want to provide high-value patient-centered care. These findings emphasize that patient decision aids are evidence-based interventions that can help support this aim,” said Dr. Trenaman. The findings also highlight an important priority: ensuring that these decision aids get into the hands of patients. “With a substantial evidence base demonstrating their effectiveness, this review highlights the importance of implementing patient decision aids into routine practice,” said Dr. Trenaman.
Washington State’s Leadership in Shared Decision-Making
Washington State has long been a global leader in supporting shared decision-making through policy. In 2007, Washington passed legislation that officially recognized shared decision making using certified high-quality patient decision aids, as a higher standard of informed consent. Washington is the only state in the U.S. that certifies patient decision aids. Since 2012, 50 patient decision aids have been certified, spanning various healthcare decisions, including maternity care, joint replacement, spine care, end-of-life care, and cancer screening.
As healthcare professionals and policymakers seek ways to enhance patient engagement and satisfaction, the incorporation of patient decision aids into standard practice represents an evidence-based strategy. Washington State’s pioneering efforts in certifying decision aids and the robust evidence from the Cochrane review reinforce the importance of integrating these tools into routine clinical practice. As healthcare systems evolve, the focus on empowering patients with the tools and information they need to make decisions aligned with their preferences will undoubtedly contribute to improved outcomes and increased patient satisfaction.
Upcoming Events
Full Calendar
News Categories
- Community Partners
- Equity, Diversity, & Inclusion
- Faculty Publications
- Faculty Research
- HSPop in the Media
- Student Research & Fieldwork
- Team Updates
To read this content please select one of the options below:
Please note you do not have access to teaching notes, stressed decision makers and use of decision aids: a literature review and conceptual model.
Information Technology & People
ISSN : 0959-3845
Article publication date: 13 November 2019
Issue publication date: 9 March 2020
Decision making is inherently stressful since the decision maker must choose between potentially conflicting alternatives with unique hazards and uncertain outcomes. Whereas decision aids such as decision support systems (DSS) can be beneficial in stressful scenarios, decision makers sometimes misuse them during decision making, leading to suboptimal outcomes. The purpose of this paper is to investigate the relationship between stress, decision making and decision aid use.
Design/methodology/approach
The authors conduct an extensive multi-disciplinary review of decision making and DSS use through the lens of stress and examine how stress, as perceived by decision makers, impacts their use or misuse of DSS even when such aids can improve decision quality. Research questions examine underlying sources of stress in managerial decision making that influence decision quality, relationships between a decision maker’s perception of stress, DSS use/misuse, and decision quality, and implications for research and practice on DSS design and capabilities.
The study presents a conceptual model that provides an integrative behavioral view of the impact of a decision maker’s perceived stress on their use of a DSS and the quality of their decisions. The authors identify critical knowledge gaps and propose a research agenda to improve decision quality and use of DSS by considering a decision maker’s perceived stress.
Originality/value
This study provides a previously unexplored view of DSS use and misuse as shaped by the decision and job stress experienced by decision makers. Through the application of four theories, the review and its findings highlight key design principles that can mitigate the negative effects of stressors on DSS use.
- Decision making
- Behaviour change
- Decision support
- Work performance
- Restrictiveness
- Automation bias
Adya, M. and Phillips-Wren, G. (2020), "Stressed decision makers and use of decision aids: a literature review and conceptual model", Information Technology & People , Vol. 33 No. 2, pp. 710-754. https://doi.org/10.1108/ITP-04-2019-0194
Emerald Publishing Limited
Copyright © 2019, Emerald Publishing Limited
Related articles
All feedback is valuable.
Please share your general feedback
Report an issue or find answers to frequently asked questions
Contact Customer Support
Jump to navigation
- Bahasa Malaysia

Patient decision aids to help people who are facing decisions about health treatment or screening
Review question
How effective/beneficial are patient decision aids for adults making decisions regarding health treatment or screening?
Key messages
- Patient decision aids are pamphlets or videos used in person or online. They clearly identify the healthcare decision to be made, provide information on options (benefits and harms), and help people clarify what is most important to them. Decision aids are designed to enhance and supplement consultation with the clinician, not replace it.
- Over 200 studies showed that patient decision aids helped adults be more involved in making health decisions by improving their knowledge and expectations of benefits and harms, and choosing an option that reflected what was most important to them.
- There were no unwanted effects for adults who used a patient decision aid.
What are patient decision aids?
Patient decision aids can help guide people making decisions when there is more than one option, including status quo (no change). They are pamphlets, videos, or web-based resources that state the decision, describe the options, and help people think about which features of the options are most important to them (which features matter most). Usual care was defined as general information, risk assessment, clinical practice guideline summaries for health consumers, placebo intervention (e.g. information on another topic), or no intervention.
What did we want to find out?
We wanted to find out if patient decision aids used by patients who are facing health treatment or screening decisions are better than the usual care for choosing an option that reflects what is most important to them. We also wanted to find out if patient decision aids were associated with any unwanted effects.
What did we do?
We updated a previous Cochrane review that was first published in 2003 and then updated in 2017. Our search included studies that compared a patient decision aid with usual care in adults who were facing health decisions for themselves or a family member. Usual care may have been general patient information or nothing. We compared and summarized the results of the studies and rated our confidence in the certainty of the evidence.
What did we find?
We found 209 studies that involved 107,698 adults. The patient decision aids focused on 71 different decisions. The common decisions were about: surgery, screening (e.g. prostate cancer, colon cancer, prenatal), genetic testing, and long-term medication treatments (e.g. insulin injections for diabetes, or statins for high cholesterol).
We are moderately confident that adults given patient decision aids were more likely to choose an option that reflected what features of the options were most important to them. Our confidence in the evidence is only moderate because the studies that provided results for our review represent only a small set of the studies evaluating patient decision aids. We are confident that when adults used patient decision aids, they had large increases in their knowledge, expectations of benefits and harms, and participation in making the decision. We are also confident that they felt better informed and were more clear about what mattered most to them. We are confident that patient decision aids did not cause any unwanted effects such as regret about the decision.
What are the limitations of the evidence?
Further research could strengthen the confidence in the evidence for choosing options that reflect which features of the options are most important to people.
How up-to-date is this evidence?
This review updates our previous review published in 2017. The evidence is up-to-date to March 2022.
Compared to usual care, across a wide variety of decisions, patient decision aids probably helped more adults reach informed values-congruent choices. They led to large increases in knowledge, accurate risk perceptions, and an active role in decision-making. Our updated review also found that patient decision aids increased patients’ feeling informed and clear about their personal values. There was no difference in decision regret between people using decision aids versus those receiving usual care. Further studies are needed to assess the impact of patient decision aids on adherence and downstream effects on cost and resource use.
Patient decision aids are interventions designed to support people making health decisions. At a minimum, patient decision aids make the decision explicit, provide evidence-based information about the options and associated benefits/harms, and help clarify personal values for features of options. This is an update of a Cochrane review that was first published in 2003 and last updated in 2017.
To assess the effects of patient decision aids in adults considering treatment or screening decisions using an integrated knowledge translation approach.
We conducted the updated search for the period of 2015 (last search date) to March 2022 in CENTRAL, MEDLINE, Embase, PsycINFO, EBSCO, and grey literature. The cumulative search covers database origins to March 2022.
We included published randomized controlled trials comparing patient decision aids to usual care. Usual care was defined as general information, risk assessment, clinical practice guideline summaries for health consumers, placebo intervention (e.g. information on another topic), or no intervention.
Two authors independently screened citations for inclusion, extracted intervention and outcome data, and assessed risk of bias using the Cochrane risk of bias tool. Primary outcomes, based on the International Patient Decision Aid Standards (IPDAS), were attributes related to the choice made (informed values-based choice congruence) and the decision-making process, such as knowledge, accurate risk perceptions, feeling informed, clear values, participation in decision-making, and adverse events. Secondary outcomes were choice, confidence in decision-making, adherence to the chosen option, preference-linked health outcomes, and impact on the healthcare system (e.g. consultation length).
We pooled results using mean differences (MDs) and risk ratios (RRs) with 95% confidence intervals (CIs), applying a random-effects model. We conducted a subgroup analysis of 105 studies that were included in the previous review version compared to those published since that update (n = 104 studies). We used Grading of Recommendations Assessment, Development, and Evaluation (GRADE) to assess the certainty of the evidence.
This update added 104 new studies for a total of 209 studies involving 107,698 participants. The patient decision aids focused on 71 different decisions. The most common decisions were about cardiovascular treatments (n = 22 studies), cancer screening (n = 17 studies colorectal, 15 prostate, 12 breast), cancer treatments (e.g. 15 breast, 11 prostate), mental health treatments (n = 10 studies), and joint replacement surgery (n = 9 studies). When assessing risk of bias in the included studies, we rated two items as mostly unclear (selective reporting: 100 studies; blinding of participants/personnel: 161 studies), due to inadequate reporting. Of the 209 included studies, 34 had at least one item rated as high risk of bias.
There was moderate-certainty evidence that patient decision aids probably increase the congruence between informed values and care choices compared to usual care (RR 1.75, 95% CI 1.44 to 2.13; 21 studies, 9377 participants).
Regarding attributes related to the decision-making process and compared to usual care, there was high-certainty evidence that patient decision aids result in improved participants' knowledge (MD 11.90/100, 95% CI 10.60 to 13.19; 107 studies, 25,492 participants), accuracy of risk perceptions (RR 1.94, 95% CI 1.61 to 2.34; 25 studies, 7796 participants), and decreased decisional conflict related to feeling uninformed (MD -10.02, 95% CI -12.31 to -7.74; 58 studies, 12,104 participants), indecision about personal values (MD -7.86, 95% CI -9.69 to -6.02; 55 studies, 11,880 participants), and proportion of people who were passive in decision-making (clinician-controlled) (RR 0.72, 95% CI 0.59 to 0.88; 21 studies, 4348 participants).
For adverse outcomes, there was high-certainty evidence that there was no difference in decision regret between the patient decision aid and usual care groups (MD -1.23, 95% CI -3.05 to 0.59; 22 studies, 3707 participants).
Of note, there was no difference in the length of consultation when patient decision aids were used in preparation for the consultation (MD -2.97 minutes, 95% CI -7.84 to 1.90; 5 studies, 420 participants). When patient decision aids were used during the consultation with the clinician, the length of consultation was 1.5 minutes longer (MD 1.50 minutes, 95% CI 0.79 to 2.20; 8 studies, 2702 participants).
We found the same direction of effect when we compared results for patient decision aid studies reported in the previous update compared to studies conducted since 2015.
- Corpus ID: 46466266
Shared decision making and decision aids - a literature review.
- J. Thistlethwaite , R. Evans , +1 author C. Heal
- Published in Australian Family Physician 1 July 2006
34 Citations
Consideration of shared decision making in nursing: a review of clinicians’ perceptions and interventions, shared decision making between registrars and patients--web based decision aids..
- Highly Influenced
Development of a program theory for shared decision-making: a realist review protocol
Acceptance of shared decision making with reference to an electronic library of decision aids (arriba-lib) and its association to decision making in patients: an evaluation study, oncology residents’ perspectives on communication skills and shared decision making, use of visual decision aids in physician–patient communication, developing a decision aid to support informed choices for newly diagnosed patients with localized prostate cancer, arriba-lib: association of an evidence-based electronic library of decision aids with communication and decision-making in patients and primary care physicians., understanding acute care nurse practitioner communication and decision-making in healthcare teams., arriba-lib: evaluation of an electronic library of decision aids in primary care physicians, related papers.
Showing 1 through 3 of 0 Related Papers
Cochrane Consumers and Communication
New review: decision aids for people facing health treatment or screening decisions.

Dawn Stacey and colleagues recently published their review. Read the plain language statement below or see the full review on the Cochrane Library.
Decision aids are interventions that support patients by making their decisions explicit, providing information about options and associated benefits/harms, and helping clarify congruence between decisions and personal values.
To assess the effects of decision aids in people facing treatment or screening decisions.
Search methods
Updated search (2012 to April 2015) in CENTRAL; MEDLINE; Embase; PsycINFO; and grey literature; includes CINAHL to September 2008.
Selection criteria
We included published randomized controlled trials comparing decision aids to usual care and/or alternative interventions. For this update, we excluded studies comparing detailed versus simple decision aids.
Data collection and analysis
Two reviewers independently screened citations for inclusion, extracted data, and assessed risk of bias. Primary outcomes, based on the International Patient Decision Aid Standards (IPDAS), were attributes related to the choice made and the decision‐making process.
Secondary outcomes were behavioural, health, and health system effects.
We pooled results using mean differences (MDs) and risk ratios (RRs), applying a random‐effects model. We conducted a subgroup analysis of studies that used the patient decision aid to prepare for the consultation and of those that used it in the consultation. We used GRADE to assess the strength of the evidence.
Main results
We included 105 studies involving 31,043 participants. This update added 18 studies and removed 28 previously included studies comparing detailed versus simple decision aids. During the 'Risk of bias' assessment, we rated two items (selective reporting and blinding of participants/personnel) as mostly unclear due to inadequate reporting. Twelve of 105 studies were at high risk of bias.
With regard to the attributes of the choice made, decision aids increased participants' knowledge (MD 13.27/100; 95% confidence interval (CI) 11.32 to 15.23; 52 studies; N = 13,316; high‐quality evidence), accuracy of risk perceptions (RR 2.10; 95% CI 1.66 to 2.66; 17 studies; N = 5096; moderate‐quality evidence), and congruency between informed values and care choices (RR 2.06; 95% CI 1.46 to 2.91; 10 studies; N = 4626; low‐quality evidence) compared to usual care.
Regarding attributes related to the decision‐making process and compared to usual care, decision aids decreased decisional conflict related to feeling uninformed (MD −9.28/100; 95% CI −12.20 to −6.36; 27 studies; N = 5707; high‐quality evidence), indecision about personal values (MD −8.81/100; 95% CI −11.99 to −5.63; 23 studies; N = 5068; high‐quality evidence), and the proportion of people who were passive in decision making (RR 0.68; 95% CI 0.55 to 0.83; 16 studies; N = 3180; moderate‐quality evidence).
Decision aids reduced the proportion of undecided participants and appeared to have a positive effect on patient‐clinician communication. Moreover, those exposed to a decision aid were either equally or more satisfied with their decision, the decision‐making process, and/or the preparation for decision making compared to usual care.
Decision aids also reduced the number of people choosing major elective invasive surgery in favour of more conservative options (RR 0.86; 95% CI 0.75 to 1.00; 18 studies; N = 3844), but this reduction reached statistical significance only after removing the study on prophylactic mastectomy for breast cancer gene carriers (RR 0.84; 95% CI 0.73 to 0.97; 17 studies; N = 3108). Compared to usual care, decision aids reduced the number of people choosing prostate‐specific antigen screening (RR 0.88; 95% CI 0.80 to 0.98; 10 studies; N = 3996) and increased those choosing to start new medications for diabetes (RR 1.65; 95% CI 1.06 to 2.56; 4 studies; N = 447). For other testing and screening choices, mostly there were no differences between decision aids and usual care.
The median effect of decision aids on length of consultation was 2.6 minutes longer (24 versus 21; 7.5% increase). The costs of the decision aid group were lower in two studies and similar to usual care in four studies. People receiving decision aids do not appear to differ from those receiving usual care in terms of anxiety, general health outcomes, and condition‐specific health outcomes. Studies did not report adverse events associated with the use of decision aids.
In subgroup analysis, we compared results for decision aids used in preparation for the consultation versus during the consultation, finding similar improvements in pooled analysis for knowledge and accurate risk perception. For other outcomes, we could not conduct formal subgroup analyses because there were too few studies in each subgroup.
Authors' conclusions
Compared to usual care across a wide variety of decision contexts, people exposed to decision aids feel more knowledgeable, better informed, and clearer about their values, and they probably have a more active role in decision making and more accurate risk perceptions. There is growing evidence that decision aids may improve values‐congruent choices. There are no adverse effects on health outcomes or satisfaction. New for this updated is evidence indicating improved knowledge and accurate risk perceptions when decision aids are used either within or in preparation for the consultation. Further research is needed on the effects on adherence with the chosen option, cost‐effectiveness, and use with lower literacy populations.
- Search Menu
- Sign in through your institution
- Advance Articles
- Editor's Choice
- Supplements
- Patient Perspectives
- Methods Corner
- Science for Patients
- Invited Commentaries
- ESC Content Collections
- Knowledge Translation
- Author Guidelines
- Instructions for reviewers
- Submission Site
- Why publish with EJCN?
- Open Access Options
- Self-Archiving Policy
- Read & Publish
- About European Journal of Cardiovascular Nursing
- About ACNAP
- About European Society of Cardiology
- ESC Publications
- Editorial Board
- Advertising & Corporate Services
- War in Ukraine
- Journals on Oxford Academic
- Books on Oxford Academic

Article Contents
Introduction, conclusions, supplementary material, acknowledgements, author contributions, data availability.
- < Previous
Patient decision aids for aortic stenosis and chronic coronary artery disease: a systematic review and meta-analysis
Conflict of interest: F.A., E.H., and D.C. received research funding in a competitive peer review process from Grow MedTech (POF000186 and POF000103) and the National Institute of Health and Care Research (NIHR204012) to develop and test a PCI PtDA called CONNECT, which is included in this review. A second author (A.B.), not involved in the development or testing of CONNECT, assessed its quality. A.-Y.C. receives honoraria and an unrestricted grant from Abbott. A.B., J.S., and H.C. have no conflicting interests.
- Article contents
- Figures & tables
- Supplementary Data
Emma Harris, Alex Benham, John Stephenson, Dwayne Conway, Aun-Yeong Chong, Helen Curtis, Felicity Astin, Patient decision aids for aortic stenosis and chronic coronary artery disease: a systematic review and meta-analysis, European Journal of Cardiovascular Nursing , Volume 23, Issue 6, August 2024, Pages 561–581, https://doi.org/10.1093/eurjcn/zvad138
- Permissions Icon Permissions
Shared decision-making is recommended for patients considering treatment options for severe aortic stenosis (AS) and chronic coronary artery disease (CAD). This review aims to systematically identify and assess patient decision aids (PtDAs) for chronic CAD and AS and evaluate the international evidence on their effectiveness for improving the quality of decision-making.
Five databases (Cochrane, CINAHL, Embase, MEDLINE, and PsycInfo), clinical trial registers, and 30 PtDA repositories/websites were searched from 2006 to March 2023. Screening, data extraction, and quality assessments were completed independently by multiple reviewers. Meta-analyses were conducted using Stata statistical software. Eleven AS and 10 CAD PtDAs were identified; seven were less than 5 years old. Over half of the PtDAs were web based and the remainder paper based. One AS and two CAD PtDAs fully/partially achieved international PtDA quality criteria. Ten studies were included in the review; four reported on the development/evaluation of AS PtDAs and six on CAD PtDAs. Most studies were conducted in the USA with White, well-educated, English-speaking participants. No studies fulfilled all quality criteria for reporting PtDA development and evaluation. Meta-analyses found that PtDAs significantly increased patient knowledge compared with ‘usual care’ (mean difference: 0.620; 95% confidence interval 0.396–0.845, P < 0.001) but did not change decisional conflict.
Patients who use PtDAs when considering treatments for AS or chronic CAD are likely to be better informed than those who do not. Existing PtDAs may not meet the needs of people with low health literacy levels as they are rarely involved in their development.
PROSPERO: CRD42021264700.

This is the first review to systematically identify and evaluate the availability, characteristics, and quality of patient decision aids for use in severe aortic stenosis and chronic coronary artery disease patient pathways.
A barrier to implementing shared decision-making for people with heart disease or aortic stenosis is the lack of high-quality, up-to-date, publicly available patient decision aids.
Existing patient decision aids are not tailored to meet the needs of people with low health literacy levels or from underserved populations.
Patient decision aids in this review improved patient knowledge, but decisional conflict scores were unchanged, possibly due to a ceiling effect.
Over the last 60 years, technological innovations have revolutionized the field of interventional cardiology. Two of the most common interventions are percutaneous coronary intervention (PCI) and transcatheter aortic valve implantation/replacement (TAVI). Over 965 000 PCIs are performed annually in the USA alone. 1 Global projections of the annual number of TAVI procedures are estimated to rise to 300 000 implants by 2025. 2 Both interventions have the potential to relieve symptoms that negatively impact quality of life 3 , 4
Patients with chronic coronary artery disease (CAD) may experience symptoms of angina. First-line treatment is medication, but if this is not effective, PCI is a treatment option to consider. 5 Patients with severe aortic stenosis (AS) also live with unpleasant symptoms associated with heart failure. Clinical guidelines indicate that a multi-disciplinary heart team should evaluate the degree of AS along with clinical and anatomical characteristics to inform their recommendations to patients about treatment options, such as TAVI or surgical aortic valve replacement (SAVR). 6
Whilst PCI and TAVI are different interventions, the decision-making processes share common features; the decision to go ahead with the treatment is considered to be ‘preference sensitive’; i.e. two or more treatment options exist but the ‘best’ treatment depends on how acceptable the patient views the potential risks and benefits of each. 7 In these situations, a process of shared decision-making (SDM) helps patients make an informed choice. 8 Accordingly, The American College of Cardiology and European Society for Cardiology recommend that SDM should take place before a patient agrees to an interventional procedure for chronic CAD or AS. 5 , 6 , 9 , 10
Shared decision-making involves a two-way discussion in which patients are informed by their doctors and nurses about what a treatment involves, the benefits and risks, and alternative options and what the outcome might be if they decided against having treatment. Importantly, SDM means that patients are encouraged to consider their unique preferences, goals, and values (i.e. what matters most to an individual about attributes of a health decision). 11 , 12 In today’s clinical practice, SDM may be difficult to achieve. Patients’ preferences and goals for treatment are not routinely discussed. 13 Moreover, patients treated with PCI often misunderstand the treatment benefits and risks and perceive their treatment as a ‘fix’. 14 , 15 Patients considering TAVI experience uncertainty about their treatment decision 16 and want to understand the risks and benefits of all potential treatment options and outcomes (e.g. TAVI, SAVR, or no intervention). 17
Patient decision aids (PtDAs) are effective interventions known to improve the quality of both the decision-making process and the choice made. 18 Evidence shows that PtDAs increase patients’ knowledge about treatments and support more accurate perceptions of associated benefits and risks. 18 However, PtDAs are not routinely used in clinical practice despite the potential benefits. 19 Some cardiologists’ do not perceive PtDAs to be of benefit to their patients. 20 Unfamiliarity and a lack of awareness of PtDAs and disagreement with the content are also factors that compromise implementation. 21
A recent meta-analysis reported that cardiology PtDAs improved two key decision outcomes: decisional conflict and patient knowledge. 22 These findings support the use of PtDAs. However, the review did not report the availability, content, and quality of the PtDAs, include PtDAs for AS, or summarize evidence on other decision-making constructs, leaving gaps in the evidence base. Accordingly, the aims of this review were to (i) identify PtDAs for chronic CAD and AS that include PCI and TAVI as treatment options and evaluate their availability, characteristics, and quality; (ii) identify and describe the quality of studies reporting on the development and evaluation of identified PtDAs; and (iii) evaluate their effectiveness on improving the quality of the decision-making process and the choice made. Findings will provide cardiology teams with an international overview of available PtDAs designed to improve the quality of SDM for chronic CAD and AS.
Review approach
Our review methods were informed by previous reviews 23 , 24 and Cochrane guidance. 25 To support the robustness of this review, the protocol was developed and registered on PROSPERO (CRD42021264700) a priori and Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines 26 implemented (see Supplementary material online , Table S1 ).
Search strategy
A search of multiple databases, trial registries, PtDA repositories, and websites was conducted, to identify eligible PtDAs and published articles that described their development or evaluation. A search strategy was developed by an information technologist (H.C.), piloted on MEDLINE (Ovid), refined, and applied to five databases in all languages: CENTRAL via the Cochrane Library, CINAHL (EBSCO), Embase, Ovid MEDLINE, and APA PsycInfo (ProQuest). Four trial registers were searched: EU clinical trials register, ClinicalTrials.gov, ISRCTN Registry, and ICTRP (WHO). Searches were limited to articles published since 1 January 2006, because the consensus on criteria for judging the quality of PtDAs was published in 2006 by the International Patient Decision Aid Standards (IPDAS) Collaboration. 27 Thirty PtDA repositories/websites were also hand searched. Searches were conducted in July 2021 and updated in March 2023. See Supplementary material online , Tables S2 – S7 for search terms and the list of PtDA repositories/websites.
Patient decision aid eligibility and selection
Patient decision aids were defined as tools designed to help facilitate SDM between patients and health professionals. 18 Patient decision aids were eligible for inclusion if they fulfilled the following criteria:
Identified as a PtDA, decision tool or an aid to support SDM in their name/title, or by the developers/authors, or listed within a PtDA repository.
Designed for patients (18+ years) with chronic CAD or AS.
Included at least two treatment options, one of which must either be PCI or TAVI.
All identified PtDAs were independently screened for inclusion by two reviewers (E.H. and A.B.). The authors, or organizations listing PtDAs not publicly available, were contacted to request a copy. Eligible PtDAs that met the criteria, but were not available in full, were included in the overview ( Table 2 ) but not in the evaluation of PtDA characteristics ( Table 2 ).
Article eligibility and selection
Search results were independently screened for inclusion by at least two reviewers (E.H. and A.B./F.A.) in three phases: title, abstract, and full-text screening. Where disagreement occurred, consensus was achieved through discussion. Articles and study reports of any design were included providing they reported on the development, user-testing, acceptability, or evaluation of eligible PtDAs. Articles reporting on ineligible PtDAs, literature reviews, and editorials were excluded.
Data extraction
Data from each included study were independently extracted by two reviewers (E.H., D.C., A.Y.C., J.S., and A.B.) into a datasheet. Characteristics from included PtDAs were extracted by one reviewer and independently checked for accuracy by a second author. Any discrepancies in data extraction were resolved by consensus. Data were synthesized into tables and presented in a narrative.
Statistical analysis
Studies evaluating the effectiveness of PtDAs were assessed for suitability and those with the same primary endpoint pooled for a meta-analysis. Due to the heterogeneity of outcome measures, only two meta-analyses were conducted on the primary interval-level outcomes of patients’ Knowledge score and Decisional Conflict score. The meta-analyses were formulated as random effects using the DerSimionian and Laird model 28 to reflect clinical and methodological heterogeneity. For both outcomes, standardized mean differences, based on post-test statistics in intervention and control groups (intervention minus control), and associated 95% confidence intervals (CIs) were measured. For the Knowledge score outcome, clinical improvement was represented by increases in reported scores. For the Decisional Conflict score outcome, clinical improvement was represented by decreases in reported scores. Forest plots were conducted for meta-analyses of both primary outcomes, reporting synthesized estimates, and associated 95% CIs, and a Z -test for the standardized mean difference. Heterogeneity statistics were also reported, including Cochran’s Q test for heterogeneity, and the I 2 statistic.
Leave-one-out sensitivity analyses were conducted on the meta-analyses of both primary outcomes to assess the robustness of the derived estimates. Each of the k -included studies was omitted in turn, and a meta-analysis was conducted based on the remaining ( k − 1) studies. Any study that was suspected of excessive influence was flagged as an influential study. Funnel plots were proposed for analyses of small-study effects for meta-analyses in which the number of identified studies reached the recommended minimum 25 but were not conducted. No sub-group analyses were identified. All analysis was conducted using Stata statistical software (Version 17 I/C). 29
Quality assessment
To support the rigour of this review, three approaches were implemented to evaluate the quality of included studies and associated PtDAs. First, the quality of PtDAs was evaluated using the six qualifying and six certification criteria of the IPDAS version four checklist, 30 which are the minimum standards for tools to be defined as a PtDA and deemed as adequate for patient use. As these criteria are designed for the evaluation of ‘full’ PtDAs, we excluded brief one- to two-page consultation/conversation aids from this assessment. Second, studies reporting an evaluation of PtDAs were assessed using the ‘Standards for UNiversal reporting of patient Decision Aid Evaluations’ (SUNDAE) checklist. 31 A modified version of this checklist was used for PtDA development studies. The IPDAS and SUNDAE checklists were independently completed by two reviewers and disagreements were resolved through discussions with a third reviewer (E.H. and A.B./F.A.). To increase the consistency of the assessments, three response options were developed: yes, partially, and no (see Supplementary material online , Tables S8 and S9 ). Third, the studies included in the meta-analyses were independently assessed by two reviewers (E.H. and F.A./J.S.) for risk of bias using either the Cochrane Risk of Bias 2 tool (RoB2 32 ) or the NHLBI Quality Assessment of Controlled Intervention Studies. 33
Figure 1 shows the search results for AS and PCI PtDAs combined. In summary, 10 studies were eligible and included in the review, which, in total, reported on the development or evaluation of 11 PtDAs. A further 10 PtDAs were identified from a trial registry record and from online PtDA repositories and relevant websites. Therefore, a total of 21 PtDAs (11 AS and 10 CAD PtDAs) were included in this review. Results for the two groups of PtDAs are presented separately by condition (A.S. and C.A.D.).

Preferred Reporting Items for Systematic Reviews and Meta-Analyses 2020 flow-diagram. 26
Patient decision aids for aortic stenosis
Availability of patient decision aids for aortic stenosis.
The search identified 11 PtDAs designed for patients with AS considering TAVI (see Table 1 for an overview). Comparative treatment options included SAVR ( n = 9) or symptom management ( n = 2). Five PtDAs included the same content but were adapted for use by different age groups (MAGIC TAVI vs. SAVR PtDAs 44–48 ). Patient decision aids were developed either in the USA ( n = 5), 34 , 38 , 39 , 41 , 43 Canada ( n = 1), 36 , 37 or by an international panel of experts ( n = 5). 44–48 All were written in English and seven were available in other languages (two in Spanish and French 39 , 41 and five in Norwegian with translation of some sections available in 12 other languages 44–48 ). Over half ( n = 8) were web-based PtDAs 34 , 36 , 38 , 44–48 and the other three were paper based. 39 , 41 , 43 Five web-based PtDAs could be converted into a printable format. 44–48 Three PtDAs were less than five years old 36 , 38 , 41 but only one was publicly available, 41 which also fully or partially achieved all 12 IPDAS quality criteria (see Quality of patient decision aids for aortic stenosis).
Overview of patient decision aids
| PtDA . | Treatment options . | Author(s) and/or developing organization . | Date developed or updated . | Country and language . | Format . | Availability . | Source of identification . |
|---|---|---|---|---|---|---|---|
| ADVICE: Navigating Aortic Valve Treatment Choices | Brennan ., Duke University | 2017 | USA, English | Web based | Not available: Website deactivated. | Literature identified via online sources | |
| Aortic Stenosis Choice (CHOICE-AS) , | Lauck . | Ongoing | Canada, English | Web based | Not currently available. PtDA development and testing study ongoing. Contact authors for access. | Online sources | |
| Aortic valve improved treatment approaches (AVITA) tool | Shared Decision-Making Resources collaborating with Edward Lifesciences | Ongoing | USA, English | Web based | Not currently available. PtDA development and pilot study ongoing. Contact authors for access. | Trial registry NCT04755426 | |
| A decision aid for treatment options for severe aortic stenosis (TAVI vs. symptom management) | American College of Cardiology | August 2017 | USA, English, Spanish, French | Eight-page booklet (pdf) | Literature | ||
| A decision aid for treatment options for severe aortic stenosis for patients deciding between TAVI and surgery | American College of Cardiology | July 2020 | USA, English, Spanish, French | Eight-page booklet (pdf) | Literature , | ||
| Severe Aortic Stenosis Decision Aid | American College of Cardiology | 2014 | USA, English | One-page pdf | and available in in published study | Literature | |
| TAVI vs. SAVR for patients with severe symptomatic aortic stenosis at low to intermediate perioperative risk: for patients above 85 years with severe symptomatic aortic stenosis, at low or intermediate perioperative risk | MAGIC Evidence Ecosystem Foundation (BMJ RapidRecs) | May 2017 | Multiple countries, English, Norwegian; partial translation into 12 other languages on website | Web based with option to create a 13-page pdf | Online sources | ||
| TAVI vs. SAVR for patients with severe symptomatic aortic stenosis at low to intermediate perioperative risk: for patients 75–85 years with severe symptomatic aortic stenosis who are at low or intermediate perioperative risk | MAGIC Evidence Ecosystem Foundation (BMJ RapidRecs) | May 2017 | Multiple countries, English, Norwegian; partial translation into 12 other languages on website | Web based with option to create a 13-page pdf | Online sources | ||
| TAVI vs. SAVR for patients with severe symptomatic aortic stenosis at low to intermediate perioperative risk: for patients aged 65 to <75 years and eligible for transfemoral TAVI or SAVR | MAGIC Evidence Ecosystem Foundation (BMJ RapidRecs) | May 2017 | Multiple countries, English, Norwegian; partial translation into 12 other languages on website | Web based with option to create a 13-page pdf | Online sources | ||
| TAVI vs. SAVR for patients with severe symptomatic aortic stenosis at low to intermediate perioperative risk: for patients aged <65 years and eligible for transfemoral TAVI or SAVR | MAGIC Evidence Ecosystem Foundation (BMJ RapidRecs) | May 2017 | Multiple countries, English, Norwegian; partial translation into 12 other languages on website | Web based with option to create a 13-page pdf | Online sources | ||
| TAVI vs. SAVR for patients with severe symptomatic aortic stenosis at low to intermediate perioperative risk who cannot undergo transfemoral TAVR but can undergo transapical approach | MAGIC Evidence Ecosystem Foundation (BMJ RapidRecs) | May 2017 | Multiple countries, English, Norwegian; partial translation into 12 other languages on website | Web-based with option to create a 13-page pdf | Online sources | ||
| Angina treatment: stents, drugs, lifestyle changes—What's best? | Mayo Clinic | May 2021 | USA, English | Web based | Online sources | ||
| Angina: treatment options, Option Grid™ | Option Grid Collaborative | 2015/16 | USA, English | Web based | Out of date: no longer available. | Literature | |
| Chest pain (stable angina) treatment options, Option Grid™ | DynaMed Decisions, EBSCO Health | December 2021. Updated when new relevant scientific evidence becomes available | USA, English | Web based with option to create a 4-page pdf | Not publicly available. Contact EBSCO Health for cost ( ). | Online sources | |
| CONNECT: COroNary aNgioplasty dECision Tool | Harris | February 2021 | UK, English | Web based | Not currently publicly available. Randomized feasibility study ongoing. Contact authors for access. | Literature | |
| Coronary artery disease: What treatment would you prefer? | Duke University Medical Center clinicians and Healthwise | 2015 | USA, English | Web based; eight-page paper version available within publication | Web version: access unknown. Paper version shown in the in published study | Literature | |
| Deciding what to do about stable angina | NHS England Vale of York Clinical Commissioning Group | January 2017 | UK, English | Nine-page pdf | Online sources | ||
| PCI Choice: Class I/II Stable Angina | Mayo Foundation for Medical Education and Research | 2012 | USA, English | Two-page pdf | Literature , | ||
| PCI Choice: Class III Stable Angina | Mayo Foundation for Medical Education and Research | 2012 | USA, English | Two-page pdf | Literature , | ||
| Should I have angioplasty for stable chest angina? | Healthwise | Updated 2022 | USA, English | Web-based with option to create a 19-page ‘printer friendly’ version | Licence required for distribution to patients or consumers. | Literature | |
| Treatment choices for stable chest discomfort | Health Dialog and Foundation for Informed Medical Decision Making | 2014 version | USA, English | Booklet (36-page paper) and DVD (20 min) | Not publicly available. Contact Health Dialog for cost. | Literature | |
| PtDA . | Treatment options . | Author(s) and/or developing organization . | Date developed or updated . | Country and language . | Format . | Availability . | Source of identification . |
|---|---|---|---|---|---|---|---|
| ADVICE: Navigating Aortic Valve Treatment Choices | Brennan ., Duke University | 2017 | USA, English | Web based | Not available: Website deactivated. | Literature identified via online sources | |
| Aortic Stenosis Choice (CHOICE-AS) , | Lauck . | Ongoing | Canada, English | Web based | Not currently available. PtDA development and testing study ongoing. Contact authors for access. | Online sources | |
| Aortic valve improved treatment approaches (AVITA) tool | Shared Decision-Making Resources collaborating with Edward Lifesciences | Ongoing | USA, English | Web based | Not currently available. PtDA development and pilot study ongoing. Contact authors for access. | Trial registry NCT04755426 | |
| A decision aid for treatment options for severe aortic stenosis (TAVI vs. symptom management) | American College of Cardiology | August 2017 | USA, English, Spanish, French | Eight-page booklet (pdf) | Literature | ||
| A decision aid for treatment options for severe aortic stenosis for patients deciding between TAVI and surgery | American College of Cardiology | July 2020 | USA, English, Spanish, French | Eight-page booklet (pdf) | Literature , | ||
| Severe Aortic Stenosis Decision Aid | American College of Cardiology | 2014 | USA, English | One-page pdf | and available in in published study | Literature | |
| TAVI vs. SAVR for patients with severe symptomatic aortic stenosis at low to intermediate perioperative risk: for patients above 85 years with severe symptomatic aortic stenosis, at low or intermediate perioperative risk | MAGIC Evidence Ecosystem Foundation (BMJ RapidRecs) | May 2017 | Multiple countries, English, Norwegian; partial translation into 12 other languages on website | Web based with option to create a 13-page pdf | Online sources | ||
| TAVI vs. SAVR for patients with severe symptomatic aortic stenosis at low to intermediate perioperative risk: for patients 75–85 years with severe symptomatic aortic stenosis who are at low or intermediate perioperative risk | MAGIC Evidence Ecosystem Foundation (BMJ RapidRecs) | May 2017 | Multiple countries, English, Norwegian; partial translation into 12 other languages on website | Web based with option to create a 13-page pdf | Online sources | ||
| TAVI vs. SAVR for patients with severe symptomatic aortic stenosis at low to intermediate perioperative risk: for patients aged 65 to <75 years and eligible for transfemoral TAVI or SAVR | MAGIC Evidence Ecosystem Foundation (BMJ RapidRecs) | May 2017 | Multiple countries, English, Norwegian; partial translation into 12 other languages on website | Web based with option to create a 13-page pdf | Online sources | ||
| TAVI vs. SAVR for patients with severe symptomatic aortic stenosis at low to intermediate perioperative risk: for patients aged <65 years and eligible for transfemoral TAVI or SAVR | MAGIC Evidence Ecosystem Foundation (BMJ RapidRecs) | May 2017 | Multiple countries, English, Norwegian; partial translation into 12 other languages on website | Web based with option to create a 13-page pdf | Online sources | ||
| TAVI vs. SAVR for patients with severe symptomatic aortic stenosis at low to intermediate perioperative risk who cannot undergo transfemoral TAVR but can undergo transapical approach | MAGIC Evidence Ecosystem Foundation (BMJ RapidRecs) | May 2017 | Multiple countries, English, Norwegian; partial translation into 12 other languages on website | Web-based with option to create a 13-page pdf | Online sources | ||
| Angina treatment: stents, drugs, lifestyle changes—What's best? | Mayo Clinic | May 2021 | USA, English | Web based | Online sources | ||
| Angina: treatment options, Option Grid™ | Option Grid Collaborative | 2015/16 | USA, English | Web based | Out of date: no longer available. | Literature | |
| Chest pain (stable angina) treatment options, Option Grid™ | DynaMed Decisions, EBSCO Health | December 2021. Updated when new relevant scientific evidence becomes available | USA, English | Web based with option to create a 4-page pdf | Not publicly available. Contact EBSCO Health for cost ( ). | Online sources | |
| CONNECT: COroNary aNgioplasty dECision Tool | Harris | February 2021 | UK, English | Web based | Not currently publicly available. Randomized feasibility study ongoing. Contact authors for access. | Literature | |
| Coronary artery disease: What treatment would you prefer? | Duke University Medical Center clinicians and Healthwise | 2015 | USA, English | Web based; eight-page paper version available within publication | Web version: access unknown. Paper version shown in the in published study | Literature | |
| Deciding what to do about stable angina | NHS England Vale of York Clinical Commissioning Group | January 2017 | UK, English | Nine-page pdf | Online sources | ||
| PCI Choice: Class I/II Stable Angina | Mayo Foundation for Medical Education and Research | 2012 | USA, English | Two-page pdf | Literature , | ||
| PCI Choice: Class III Stable Angina | Mayo Foundation for Medical Education and Research | 2012 | USA, English | Two-page pdf | Literature , | ||
| Should I have angioplasty for stable chest angina? | Healthwise | Updated 2022 | USA, English | Web-based with option to create a 19-page ‘printer friendly’ version | Licence required for distribution to patients or consumers. | Literature | |
| Treatment choices for stable chest discomfort | Health Dialog and Foundation for Informed Medical Decision Making | 2014 version | USA, English | Booklet (36-page paper) and DVD (20 min) | Not publicly available. Contact Health Dialog for cost. | Literature | |
a https://sharedcardiology.org .
b European Society of Cardiology Website https://www.escardio.org/ .
c The Ottawa Hospital Research Institute Decision Aid Library Inventory https://decisionaid.ohri.ca/index.html .
d EBSCO Health care https://www.ebsco.com/health-care/products/my-health-decisions .
e Vale of York NHS https://www.valeofyorkccg.nhs.uk/rss/home/patient-decision-making/shared-decision-making/ .
Characteristics of patient decision aids for aortic stenosis
The characteristics of eight PtDAs for AS were evaluated ( Table 2 ). 39 , 41 , 43–48 The remaining three were unavailable for evaluation due to website deactivation 34 or ongoing development. 36 , 38
Characteristics of patient decision aids
| PtDA . | Format and delivery . | Design and development . | EVC method . | Tx preference indication . | Other interaction . | Risk/benefits presentation . | Patient stories . | No. of IPDAS criteria achieved . |
|---|---|---|---|---|---|---|---|---|
| A decision aid for treatment options for severe aortic stenosis (TAVI vs. Symptom Management) | Paper booklet reviewed by patient pre-consultation | Colour text, graphics, text boxes, photos of people, images to explain disease and procedure. 15-min video on website. Development not described. | Four questions with open-text responses about hopes, concerns, questions for HCPs and family | None | — | Side-by-side list and icon arrays (100 heart icons); natural frequencies (denominator: 100); positive and negative framing | Two scenarios. Patient’s Tx choice shown | Fully: 11 Partially: 0 Not met: 1 |
| A decision aid for treatment options for severe aortic stenosis for patients deciding between TAVI and surgery | Paper booklet reviewed by patient pre-consultation | Colour text, graphics, text boxes, photos of people, images to explain disease and procedure. 18.5-min video on website. Development not described. | Four questions with open-text responses about hopes, concerns, questions for HCPs and family | None | — | Side-by-side list and icon arrays (10 people icons); natural frequencies (denominator: 10 and 100); mostly negative framing used; positive and negative used for survival | Two scenarios. Patient’s Tx choice shown | Fully: 11 Partially: 1 Not met: 0 |
| Severe Aortic Stenosis Decision Aid | Brief one-page paper ‘Encounter PtDA’ reviewed during consultation with HCP | Colour text, text boxes, graphs Development briefly described | Conversation guide with 1 question asking the patient ‘What matters most to you?’ | Open-text response to indicate patient and HCP’s shared decision | — | Side-by-side list, icon arrays (100 circles) and line graphs; positive and negative framing | None | N/A |
| TAVI vs. SAVR for patients with severe symptomatic aortic stenosis at low to intermediate perioperative risk (5 versions for different age groups ) | Web-based interactive ‘Encounter PtDA’ reviewed during consultation with HCP | Text, pop-up boxes, black/white icons Clinical content review described on website. Option to download as pdf. | None | None | Web version only: HCP navigates between sections to guide discussion and explore outcomes the patient wants to discuss | Icon arrays (1000 people icons); side-by-side natural frequencies (denominator: 1000); mix of positive or negative framing | None | Fully: 8 Partially: 3 Not met: 1 |
| Angina treatment: stents, drugs, lifestyle changes—What's best? | Website. Delivery not specified | Text, colour image to explain procedure. Development not described. | None | None | — | Only states one risk (blockage re-forming). Likelihood not provided. | None | Fully: 5 Partially: 2 Not met: 5 |
| CONNECT: COroNary aNgioplasty dECision Tool , | Web-based reviewed by patient pre-consultation. Personalised summary to be shared with HCP during consultation | Text, drop-down boxes, pop-up boxes, tables, colour icons, colour diagrams to explain disease and procedure, multiple short animated videos, photos of people. Development fully described. | Open-text box for patient to add the top 3 things that matter most to them when considering their Tx options | Multiple-choice question with ‘not sure’ as an option. A smiley face 5-point Likert scale to indicate level of certainty with choice | Patient input: navigation between sections; six-item multiple-choice Angina Symptom Evaluation Questionnaire; Open-text box to add worries or questions. Generates personal summary of answers. | Side-by-side comparison table; icon arrays (1000 people icons for PCI risks, 100 people for benefits of both options); natural frequencies (denominator: 1000 and 5000); positive and negative framing | Text and audio quotes from 5 fictional patients. Tx choice not shown. | Fully: 12 Partially: 0 Not met: 0 |
| Coronary artery disease: What treatment would you prefer? (paper version only) | Web and paper reviewed by patient on the day of diagnostic angiogram | Paper version: Text, colour graphics, table, pictures and icons, colour diagrams to explain disease and procedures. Development described briefly. | None | One question asking patient to record preferred Tx | — | Side-by-side lists; icon arrays (100 people icons); natural frequencies (denominator: 1000); negative framing | None | Fully: 9 Partially: 0 Not met: 3 |
| Deciding what to do about stable angina | Paper based reviewed by patient pre-consultation or with HCP during consultation | Text, diagram, tables. Development not described. | None | None | Six questions for the patient to consider (no space for patient answers) | Side-by-side comparison table; positively framed natural frequencies for symptom improvement for PCI/CABG option only (denominator: 100); negatively framed natural frequencies (denominator: 100) for medicines option; descriptive words for PCI and CABG (small, low, and higher) | None | Fully: 9 Partially: 1 Not met: 2 |
| PCI choice (two versions for either Class I/II or Class III Stable Angina , ) | Brief two-page paper ‘Encounter PtDA’ reviewed during consultation with HCP | Colour text, text boxes, colour icons. Development fully described. | None | Two questions asking for preferred Tx | None | Side-by-side icon arrays (100 circles icons); natural frequencies (denominator: 100) with positive and negative framing | None | N/A |
| Should I have angioplasty for stable chest angina? | Web-based pre-consultation. Delivery determined by distributor. In publication, the link to the PtDA website was e-mailed to patients’ pre-consultation. | Web: Text, drop-down boxes, pop-up boxes, tables, colour diagrams to explain procedure with real angiogram X-ray image. Clinical content review described on website. Option to download as pdf. | Rating scales: Four 7-point ‘importance’ Likert scales for three pre-set attributes and 1 open-box for patient to add other important attributes/values. | Two 7-point Likert scales to indicate preferred Tx and level of certainty with choice | Patient input: navigation between sections; three-item yes/no knowledge test; 3 yes/no questions about support and understanding, open-text box to add worries or questions. Generates personal summary of answers. | Side-by-side list; icon arrays (100 people icons); side-by-side natural frequencies (denominator: 100) with positive and negative framing for benefits; negative framing for PCI risks | Quotes from four fictional patients. Tx choice shown. | Fully: 9 Partially: 3 Not met: 0 |
| PtDA . | Format and delivery . | Design and development . | EVC method . | Tx preference indication . | Other interaction . | Risk/benefits presentation . | Patient stories . | No. of IPDAS criteria achieved . |
|---|---|---|---|---|---|---|---|---|
| A decision aid for treatment options for severe aortic stenosis (TAVI vs. Symptom Management) | Paper booklet reviewed by patient pre-consultation | Colour text, graphics, text boxes, photos of people, images to explain disease and procedure. 15-min video on website. Development not described. | Four questions with open-text responses about hopes, concerns, questions for HCPs and family | None | — | Side-by-side list and icon arrays (100 heart icons); natural frequencies (denominator: 100); positive and negative framing | Two scenarios. Patient’s Tx choice shown | Fully: 11 Partially: 0 Not met: 1 |
| A decision aid for treatment options for severe aortic stenosis for patients deciding between TAVI and surgery | Paper booklet reviewed by patient pre-consultation | Colour text, graphics, text boxes, photos of people, images to explain disease and procedure. 18.5-min video on website. Development not described. | Four questions with open-text responses about hopes, concerns, questions for HCPs and family | None | — | Side-by-side list and icon arrays (10 people icons); natural frequencies (denominator: 10 and 100); mostly negative framing used; positive and negative used for survival | Two scenarios. Patient’s Tx choice shown | Fully: 11 Partially: 1 Not met: 0 |
| Severe Aortic Stenosis Decision Aid | Brief one-page paper ‘Encounter PtDA’ reviewed during consultation with HCP | Colour text, text boxes, graphs Development briefly described | Conversation guide with 1 question asking the patient ‘What matters most to you?’ | Open-text response to indicate patient and HCP’s shared decision | — | Side-by-side list, icon arrays (100 circles) and line graphs; positive and negative framing | None | N/A |
| TAVI vs. SAVR for patients with severe symptomatic aortic stenosis at low to intermediate perioperative risk (5 versions for different age groups ) | Web-based interactive ‘Encounter PtDA’ reviewed during consultation with HCP | Text, pop-up boxes, black/white icons Clinical content review described on website. Option to download as pdf. | None | None | Web version only: HCP navigates between sections to guide discussion and explore outcomes the patient wants to discuss | Icon arrays (1000 people icons); side-by-side natural frequencies (denominator: 1000); mix of positive or negative framing | None | Fully: 8 Partially: 3 Not met: 1 |
| Angina treatment: stents, drugs, lifestyle changes—What's best? | Website. Delivery not specified | Text, colour image to explain procedure. Development not described. | None | None | — | Only states one risk (blockage re-forming). Likelihood not provided. | None | Fully: 5 Partially: 2 Not met: 5 |
| CONNECT: COroNary aNgioplasty dECision Tool , | Web-based reviewed by patient pre-consultation. Personalised summary to be shared with HCP during consultation | Text, drop-down boxes, pop-up boxes, tables, colour icons, colour diagrams to explain disease and procedure, multiple short animated videos, photos of people. Development fully described. | Open-text box for patient to add the top 3 things that matter most to them when considering their Tx options | Multiple-choice question with ‘not sure’ as an option. A smiley face 5-point Likert scale to indicate level of certainty with choice | Patient input: navigation between sections; six-item multiple-choice Angina Symptom Evaluation Questionnaire; Open-text box to add worries or questions. Generates personal summary of answers. | Side-by-side comparison table; icon arrays (1000 people icons for PCI risks, 100 people for benefits of both options); natural frequencies (denominator: 1000 and 5000); positive and negative framing | Text and audio quotes from 5 fictional patients. Tx choice not shown. | Fully: 12 Partially: 0 Not met: 0 |
| Coronary artery disease: What treatment would you prefer? (paper version only) | Web and paper reviewed by patient on the day of diagnostic angiogram | Paper version: Text, colour graphics, table, pictures and icons, colour diagrams to explain disease and procedures. Development described briefly. | None | One question asking patient to record preferred Tx | — | Side-by-side lists; icon arrays (100 people icons); natural frequencies (denominator: 1000); negative framing | None | Fully: 9 Partially: 0 Not met: 3 |
| Deciding what to do about stable angina | Paper based reviewed by patient pre-consultation or with HCP during consultation | Text, diagram, tables. Development not described. | None | None | Six questions for the patient to consider (no space for patient answers) | Side-by-side comparison table; positively framed natural frequencies for symptom improvement for PCI/CABG option only (denominator: 100); negatively framed natural frequencies (denominator: 100) for medicines option; descriptive words for PCI and CABG (small, low, and higher) | None | Fully: 9 Partially: 1 Not met: 2 |
| PCI choice (two versions for either Class I/II or Class III Stable Angina , ) | Brief two-page paper ‘Encounter PtDA’ reviewed during consultation with HCP | Colour text, text boxes, colour icons. Development fully described. | None | Two questions asking for preferred Tx | None | Side-by-side icon arrays (100 circles icons); natural frequencies (denominator: 100) with positive and negative framing | None | N/A |
| Should I have angioplasty for stable chest angina? | Web-based pre-consultation. Delivery determined by distributor. In publication, the link to the PtDA website was e-mailed to patients’ pre-consultation. | Web: Text, drop-down boxes, pop-up boxes, tables, colour diagrams to explain procedure with real angiogram X-ray image. Clinical content review described on website. Option to download as pdf. | Rating scales: Four 7-point ‘importance’ Likert scales for three pre-set attributes and 1 open-box for patient to add other important attributes/values. | Two 7-point Likert scales to indicate preferred Tx and level of certainty with choice | Patient input: navigation between sections; three-item yes/no knowledge test; 3 yes/no questions about support and understanding, open-text box to add worries or questions. Generates personal summary of answers. | Side-by-side list; icon arrays (100 people icons); side-by-side natural frequencies (denominator: 100) with positive and negative framing for benefits; negative framing for PCI risks | Quotes from four fictional patients. Tx choice shown. | Fully: 9 Partially: 3 Not met: 0 |
EVC, explicit values clarification; HCP, healthcare professional; Tx, treatment
a Only paper version evaluated, web version unavailable.
Two types of PtDAs were identified: a PtDA booklet (eight pages) to be reviewed by the patient at home 39 , 41 and an ‘encounter PtDA’ (paper or web-based) to be used during the consultation with a health professional. 43–48 The type and presentation of information varied between PtDAs. One ‘encounter PtDA’ presented information about the risks and benefits of treatment options on a single page, 43 whereas the other ‘encounter PtDAs’ were web based and required health professionals to navigate between different sections to present the information. 44–48 All PtDAs included icon arrays to present the risks and benefits of treatment options. Patient stories were only included in the two booklet PtDAs. 39 , 41 Three PtDAs incorporated an explicit values clarification method 39 , 41 , 43 (i.e. determining what matters to patients about a given health decision by using an approach that requires interaction 12 ). The method in the two booklet PtDAs invited patients to write their hopes and concerns for the treatment options and any questions for their doctor and family. 39 , 41 The one-page ‘encounter PtDA’ invited patients to verbally respond to the question during a consultation, about what was important to them about their treatment. 43 This was the only PtDA to invite patients to indicate their preferred treatment. The readability score was not reported for any PtDA. Two PtDAs did not report their development method. 39 , 41
Quality of patient decision aids for aortic stenosis
Seven PtDAs 39 , 41 , 44–48 were included for quality appraisal using the recommended IPDAS checklist (‘encounter PtDAs’ were excluded 43 ). Results are summarized in Table 2 (full evaluation in Supplementary material online , Table S10 ). To ‘qualify’ as a PtDA, six IPDAS criteria need to be achieved; only the two booklet PtDAs fulfilled these. 39 , 41 In total, the PtDAs fulfilled between 67% and 92% (median 67%) of all 12 IPDAS criteria. Two IPDAS criteria were not achieved by all PtDAs: ‘describes the condition related to the decision’ and ‘the level of uncertainty around outcome probabilities’ (i.e. the likelihood of an adverse or positive outcome occurring following treatment).
Patient decision aids for chronic coronary artery disease
Availability of patient decision aids for coronary artery disease.
Ten PtDAs designed for patients with chronic CAD considering PCI were identified ( Table 1 ). The comparative treatment options presented were medical therapy ( n = 10), lifestyle changes ( n = 4), and coronary artery bypass graft (CABG) surgery ( n = 4). The two ‘PCI Choice’ PtDAs 58 , 61 included the same content but adapted the risks/benefits probabilities for either Class I/II or Class III stable angina. Eight PtDAs were developed in the USA 49 , 50 , 52 , 55 , 58 , 61 , 62 , 64 and two in the UK, 53 , 57 and all were only available in English. Six were web-based PtDAs 49 , 50 , 52 , 53 , 55 , 62 and four were paper based 57 , 58 , 61 , 64 (one also included a 20-min DVD 64 ). One web-based PtDA had a paper-based version 55 and two others could be converted into a printable format. 52 , 62 Four PtDAs were less than five years old 49 , 52 , 53 , 62 but only one was publicly available. 49 This PtDA 49 fulfilled only five of the 12 IPDAS criteria.
Characteristics of patient decision aids for coronary artery disease
The characteristics of seven PtDAs for chronic CAD were evaluated ( Table 2 ). 49 , 53 , 55 , 57 , 58 , 61 , 62 The remaining three were unavailable for evaluation. 50 , 52 , 64
The type of PtDA, approach, and time point of use in the patient journey varied. Two were short paper-based ‘encounter PtDAs’ (PCI Choice 58 , 61 ) to be used by the doctor with the patient in a consultation prior to diagnostic cardiac catheterization. Three web-based PtDAs 53 , 62 (one had a paper version option 55 ) could be reviewed by patients either at home or whilst in hospital before the procedure. One paper-based PtDA could be used either pre-consultation or during the consultation. 57 Details about the delivery of one web-based PtDA were absent. 49 The design of PtDAs varied from a basic table comparing treatments with the use of multi-media to explain health conditions, treatment options, and procedures. Treatment risks and benefits were presented using a wide range of approaches. All but two 49 , 57 included icon arrays to convey the likelihood of risks and benefits. One PtDA 49 omitted the major risks associated with PCI. Patient stories/scenarios were included in two PtDAs. 53 , 62 Two PtDAs included explicit value clarification methods: a rating scale 62 and completion of questions about what matters to them and their concerns. 53 Five PtDAs invited patients to indicate their preferred treatment. 53 , 55 , 58 , 61 , 62 A personalized summary of patients’ responses could be generated in two web-based PtDAs. 53 , 62 The readability level was not stated within any PtDA, although associated publications for two PtDAs reported the target reading age as eighth grade (age 13–14 years). 54 , 56 Development information was published, in varying detail, for some PtDAs, 53 , 55 , 58 , 61 two omitted this information, 49 , 57 whilst brief details about the development of clinical content were described for the remainder on the developers’ websites. 62
Quality of patient decision aids for coronary artery disease
Five PtDAs 49 , 53 , 55 , 57 , 62 were included for quality appraisal (two ‘encounter PtDAs’ were excluded 58 , 61 ; Table 2 ). Three PtDAs 53 , 55 , 62 completely fulfilled the six IPDAS ‘qualify’ criteria (see Supplementary material online , Table S10 ). In total, the five PtDAs fulfilled between 42% and 100% (median 75%) of all 12 IPDAS criteria. Two PtDAs 53 , 62 fully or partially achieved all 12 IPDAS criteria but are not currently publicly available to patients. The IPDAS criteria least fulfilled across the PtDAs were ‘providing information about the funding source’, ‘the updated policy’, and ‘the level of uncertainty around outcome probabilities’.
Overview of included studies
Table 3 provides an overview of the 10 studies included in the review (full details in Supplementary material online , Table S1 ). One study was conducted in the UK 54 and the remainder in the USA. Three reported on PtDA development and acceptability testing, 35 , 54 , 59 and seven evaluated PtDA effectiveness in either an RCT 42 , 60 or a quasi-experimental design. 20 , 40 , 51 , 56 , 63
Overview of studies
| Study details . | Study design . | Methods, sample, and setting . | Results . | No. of SUNDAE items met . |
|---|---|---|---|---|
| treatment options | ||||
| Brennan ., 2020, USA | Multi-methods development study of risk calculator and PtDA for patients with AS ( ) | Setting: Duke University Medical Center 1) Development of risk calculator: Patient survey (SAVR = 10; TAVR = 10); registry data review and questionnaire by 3 caregivers and 5 patients to identify patient characteristics to include in risk models. 2) Feedback on risk calculator: 4 rounds of semi-structured interviews with 6 TAVR and SAVR patients and caregivers. 3) SDM education resource development: multiple teleconference calls with a multi-disciplinary team including 7 patients and 3 caregivers to determine content. 4) Feedback on PtDA: Review by patient and caregiver stakeholders and semi-structured interviews with 6 patients scheduled for TAVR. | = 817. | Fully: 13 Partially: 3 Not met: 2 N/a: 8 |
| Coylewright . 2020, USA | Single-centre non-randomized pre-test post-test pilot study with 3 patient groups: UC (no PtDA); cardiologist’s 1st use of PtDA ( ; ) cardiologist’s 5th use of PtDA | Setting: 2 TAVR centres in Northern New England 35 patients (56% female) with severe AS, at high or prohibitive surgical risk, for whom HCPs agree potential equipoise for TAVR and SAVR. UC: Each cardiologist ( = 4) or pair ( = 1) audio recorded a consultation without PtDA with 5 patients each (25 total). Patients’ mean (SD) age: 85 (7.5) years; 75% achieved high-school education or greater. 1st use of PtDA: Each cardiologist/pair used the PtDA with 1 patient (5 total). Patients’ mean (SD) age: 82 (10.5) years; 100% achieved high-school education or greater (1 missing response). 5th use of PtDA: Each cardiologist/pair’s 5th time of using the PtDA with a patient ( = 5). Patients’ mean (SD) age : 93 (2.7) years; 80% achieved high-school education or greater. | Full: 19 Partial: 4 No: 3 N/a: 0 | |
| Einfeld, 2020, USA | Single-centre uncontrolled pre-post intervention (peer support and use of PtDAs in patients considering TAVR) pilot study with 1 patient group 2 PtDAs: and | Setting: Community hospital in Pacific Northwest Patients with AS ( = 12; 63–89 years; 42% Female) eligible for TAVR participated in peer-support (Mended Hearts programme). TAVR PtDAs integrated into UC consultations. | Full: 19 Partial: 3 Not: 4 N/a: 0 | |
| ., 2022, USA | Single-centre pilot 1:1 RCT (PtDA vs. UC) of a PtDA delivered to patients with AS considering TAVR or SAVR PtDA: Comparator: UC in-clinic discussion of treatment options, risks, and benefits, and an animation of the TAVR procedure. No written materials. | Setting: Massachusetts General Hospital, USA Patients ( = 60, 100% White) with mild or moderate AS being assessed for either TAVR or surgical SAVR were randomized to PtDA or UC group. PtDA ( = 31): mean age 74 (SD 6) years; 39% female; 89% achieved college education or greater. UC ( = 28): mean age 71 (SD 8) years; 25% female; 75% achieved college education or greater. | 68% reported reviewing all the PtDA | Full: 19 Partial: 2 Not: 4 N/a: 1 |
| treatment options | ||||
| Coylewright ., 2012, USA | Multi-phase development and single-centre acceptability study of PtDA ( , ) for patients with stable CAD facing treatment with either OMT or PCI + OMT | Setting: Mayo clinic 1) Evidence review and synthesis. 3) Prototype PtDA developed by 2 HCPs plus designer. 4) Tested by Diabetes Research Advisory Group (15–20 community members with DM), and Cardiovascular Patient and Family Advisory Council (over 25 patients and family members) to develop first prototype. 5) Observed use in clinical setting with 25 patients. Revised PtDA over 1–2 weeks after each clinical observation. | Fully: 13 Partially:5 Not met: 0 N/a: 8 | |
| ., 2016, USA | Single-centre, randomized controlled (1:1) trial of PtDA ( , ) vs. UC (no PtDA) | Setting: Mayo clinic 124 Patients with stable CAD considering OMT +/− PCI treatment randomized to PtDA or UC group. PtDA ( = 65): mean age 69 (SD 10.9) years; 28% female; 100% White; 65% achieved college education or greater UC ( = 59): mean age 68 (SD 10.2) years; 25% female; 98% White; 71% achieved college education or greater. | Full: 18 Partial: 5 No: 3 N/a: 0 | |
| ., 2019, USA | Two-part study: A) Single-centre prospective non-randomized controlled pre-post-test study of PtDA ( ) vs. UC (no PtDA, no treatment preferences) B) Pilot cluster randomized study embedded within above study | Setting: Duke University Hospital. A) 203 patients with chest pain, angina (acute and chronic) or NSTEMI, referred for diagnostic coronary angiography and considering treatment with either medical therapy, PCI or CABG, non-randomized to PtDA or UC group. UC ( = 100): median age (IQR) 64 (56–70) years; 34% female; 76% White; 63% achieved college education or greater; Health literacy mean (SD) 2 (2.6). PtDA ( = 103): median age (IQR) 63 (55–72) years; 43% female; 71% White; 71% achieved college education or greater; Health literacy mean (SD) 1.5 (2.1). B) 103 patients in PtDA group randomized 50:53 to preference group (cardiologist received patients’ treatment preferences) or control group (preferences not shared). | Full: 13 Partial: 8 Not: 5 N/a: 0 | |
| Harris ., 2022, UK | Multi-phase, multi-centre development and acceptability testing of a PtDA for people with stable angina considering elective coronary angioplasty treatment ( ) | Setting: 2 District General Hospitals in Northern England. 34 patients and 29 HCPs in total involved in various stages 1) Steering Group convened, evidence review, and 3 co-design workshops with 4 cardiologists, 9 nurses, and 9 members of heart support groups. 2) Alpha-testing of prototype 1 (cognitive interviews and acceptability questionnaire) with 9 HCPs and 6 patients, 1 patient/partner dyad in non-clinical settings. Patient sample: mean age 63 (SD 11) years; 29% female; 85% achieved college education; 71% had adequate HL. 3) PtDA refined and prototype 2 developed following consultations with 10 service users, 7 HCPs and the Steering Group. Feedback on prototype 2 collated from 9 new volunteers from community heart support groups, 1 Steering Group lay member, and 2 consultant cardiologists. | Full: 16 Partial: 2 Not: 0 N/a: 8 | |
| Hinsberg ., 2018, USA | Single-centre randomized comparator pilot trial to compare effects of two PtDAs for stable angina. DVD/booklet PtDA: Web-based PtDA: | Setting: Massachusetts General Hospital Heart Centre Patients ( = 28) who had recently made decisions about treatment of stable CAD were randomized to DVD/paper booklet PtDA or web-based PtDA. DVD/booklet PtDA ( = 15): mean age 73 (SD 11.6) years; 60% female; 100% White; 80% achieved college education or greater. Web-based PtDA ( = 13): mean age 67 (SD 10.62) years; 23% female; 92% White; 54% achieved college education or greater. | = 0.05) | Full: 19 Partial: 4 Not: 2 N/a: 1 |
| Scalia ., 2018, USA | Cross-sectional observational study to evaluate whether Option Grid PtDAs change treatment preferences and which items of the PtDA are most important to users PtDA: | Audit data collected from users of Option Grid PtDAs who had an account on the Option Grid website, over a 19-month period (June 2015 onwards). User responses in the PtDAs were collected from the top 5 most-used PtDAs. The Angina PtDA was accessed and fully completed by 88 users (47% female; 11% Hispanic, 46% not Hispanic, 43% ethnicity not stated; age range: 11% 20–30 years, 16% 31–40 years, 18% 41–50 years, 17% 51–60 years, 10% ≥60 years, 27% not stated). | For Angina treatment options: no significant preference shift between medical management and stenting; = 0.200. | Full: 16 Partial: 6 Not: 2 N/a: 2 |
| Study details . | Study design . | Methods, sample, and setting . | Results . | No. of SUNDAE items met . |
|---|---|---|---|---|
| treatment options | ||||
| Brennan ., 2020, USA | Multi-methods development study of risk calculator and PtDA for patients with AS ( ) | Setting: Duke University Medical Center 1) Development of risk calculator: Patient survey (SAVR = 10; TAVR = 10); registry data review and questionnaire by 3 caregivers and 5 patients to identify patient characteristics to include in risk models. 2) Feedback on risk calculator: 4 rounds of semi-structured interviews with 6 TAVR and SAVR patients and caregivers. 3) SDM education resource development: multiple teleconference calls with a multi-disciplinary team including 7 patients and 3 caregivers to determine content. 4) Feedback on PtDA: Review by patient and caregiver stakeholders and semi-structured interviews with 6 patients scheduled for TAVR. | = 817. | Fully: 13 Partially: 3 Not met: 2 N/a: 8 |
| Coylewright . 2020, USA | Single-centre non-randomized pre-test post-test pilot study with 3 patient groups: UC (no PtDA); cardiologist’s 1st use of PtDA ( ; ) cardiologist’s 5th use of PtDA | Setting: 2 TAVR centres in Northern New England 35 patients (56% female) with severe AS, at high or prohibitive surgical risk, for whom HCPs agree potential equipoise for TAVR and SAVR. UC: Each cardiologist ( = 4) or pair ( = 1) audio recorded a consultation without PtDA with 5 patients each (25 total). Patients’ mean (SD) age: 85 (7.5) years; 75% achieved high-school education or greater. 1st use of PtDA: Each cardiologist/pair used the PtDA with 1 patient (5 total). Patients’ mean (SD) age: 82 (10.5) years; 100% achieved high-school education or greater (1 missing response). 5th use of PtDA: Each cardiologist/pair’s 5th time of using the PtDA with a patient ( = 5). Patients’ mean (SD) age : 93 (2.7) years; 80% achieved high-school education or greater. | Full: 19 Partial: 4 No: 3 N/a: 0 | |
| Einfeld, 2020, USA | Single-centre uncontrolled pre-post intervention (peer support and use of PtDAs in patients considering TAVR) pilot study with 1 patient group 2 PtDAs: and | Setting: Community hospital in Pacific Northwest Patients with AS ( = 12; 63–89 years; 42% Female) eligible for TAVR participated in peer-support (Mended Hearts programme). TAVR PtDAs integrated into UC consultations. | Full: 19 Partial: 3 Not: 4 N/a: 0 | |
| ., 2022, USA | Single-centre pilot 1:1 RCT (PtDA vs. UC) of a PtDA delivered to patients with AS considering TAVR or SAVR PtDA: Comparator: UC in-clinic discussion of treatment options, risks, and benefits, and an animation of the TAVR procedure. No written materials. | Setting: Massachusetts General Hospital, USA Patients ( = 60, 100% White) with mild or moderate AS being assessed for either TAVR or surgical SAVR were randomized to PtDA or UC group. PtDA ( = 31): mean age 74 (SD 6) years; 39% female; 89% achieved college education or greater. UC ( = 28): mean age 71 (SD 8) years; 25% female; 75% achieved college education or greater. | 68% reported reviewing all the PtDA | Full: 19 Partial: 2 Not: 4 N/a: 1 |
| treatment options | ||||
| Coylewright ., 2012, USA | Multi-phase development and single-centre acceptability study of PtDA ( , ) for patients with stable CAD facing treatment with either OMT or PCI + OMT | Setting: Mayo clinic 1) Evidence review and synthesis. 3) Prototype PtDA developed by 2 HCPs plus designer. 4) Tested by Diabetes Research Advisory Group (15–20 community members with DM), and Cardiovascular Patient and Family Advisory Council (over 25 patients and family members) to develop first prototype. 5) Observed use in clinical setting with 25 patients. Revised PtDA over 1–2 weeks after each clinical observation. | Fully: 13 Partially:5 Not met: 0 N/a: 8 | |
| ., 2016, USA | Single-centre, randomized controlled (1:1) trial of PtDA ( , ) vs. UC (no PtDA) | Setting: Mayo clinic 124 Patients with stable CAD considering OMT +/− PCI treatment randomized to PtDA or UC group. PtDA ( = 65): mean age 69 (SD 10.9) years; 28% female; 100% White; 65% achieved college education or greater UC ( = 59): mean age 68 (SD 10.2) years; 25% female; 98% White; 71% achieved college education or greater. | Full: 18 Partial: 5 No: 3 N/a: 0 | |
| ., 2019, USA | Two-part study: A) Single-centre prospective non-randomized controlled pre-post-test study of PtDA ( ) vs. UC (no PtDA, no treatment preferences) B) Pilot cluster randomized study embedded within above study | Setting: Duke University Hospital. A) 203 patients with chest pain, angina (acute and chronic) or NSTEMI, referred for diagnostic coronary angiography and considering treatment with either medical therapy, PCI or CABG, non-randomized to PtDA or UC group. UC ( = 100): median age (IQR) 64 (56–70) years; 34% female; 76% White; 63% achieved college education or greater; Health literacy mean (SD) 2 (2.6). PtDA ( = 103): median age (IQR) 63 (55–72) years; 43% female; 71% White; 71% achieved college education or greater; Health literacy mean (SD) 1.5 (2.1). B) 103 patients in PtDA group randomized 50:53 to preference group (cardiologist received patients’ treatment preferences) or control group (preferences not shared). | Full: 13 Partial: 8 Not: 5 N/a: 0 | |
| Harris ., 2022, UK | Multi-phase, multi-centre development and acceptability testing of a PtDA for people with stable angina considering elective coronary angioplasty treatment ( ) | Setting: 2 District General Hospitals in Northern England. 34 patients and 29 HCPs in total involved in various stages 1) Steering Group convened, evidence review, and 3 co-design workshops with 4 cardiologists, 9 nurses, and 9 members of heart support groups. 2) Alpha-testing of prototype 1 (cognitive interviews and acceptability questionnaire) with 9 HCPs and 6 patients, 1 patient/partner dyad in non-clinical settings. Patient sample: mean age 63 (SD 11) years; 29% female; 85% achieved college education; 71% had adequate HL. 3) PtDA refined and prototype 2 developed following consultations with 10 service users, 7 HCPs and the Steering Group. Feedback on prototype 2 collated from 9 new volunteers from community heart support groups, 1 Steering Group lay member, and 2 consultant cardiologists. | Full: 16 Partial: 2 Not: 0 N/a: 8 | |
| Hinsberg ., 2018, USA | Single-centre randomized comparator pilot trial to compare effects of two PtDAs for stable angina. DVD/booklet PtDA: Web-based PtDA: | Setting: Massachusetts General Hospital Heart Centre Patients ( = 28) who had recently made decisions about treatment of stable CAD were randomized to DVD/paper booklet PtDA or web-based PtDA. DVD/booklet PtDA ( = 15): mean age 73 (SD 11.6) years; 60% female; 100% White; 80% achieved college education or greater. Web-based PtDA ( = 13): mean age 67 (SD 10.62) years; 23% female; 92% White; 54% achieved college education or greater. | = 0.05) | Full: 19 Partial: 4 Not: 2 N/a: 1 |
| Scalia ., 2018, USA | Cross-sectional observational study to evaluate whether Option Grid PtDAs change treatment preferences and which items of the PtDA are most important to users PtDA: | Audit data collected from users of Option Grid PtDAs who had an account on the Option Grid website, over a 19-month period (June 2015 onwards). User responses in the PtDAs were collected from the top 5 most-used PtDAs. The Angina PtDA was accessed and fully completed by 88 users (47% female; 11% Hispanic, 46% not Hispanic, 43% ethnicity not stated; age range: 11% 20–30 years, 16% 31–40 years, 18% 41–50 years, 17% 51–60 years, 10% ≥60 years, 27% not stated). | For Angina treatment options: no significant preference shift between medical management and stenting; = 0.200. | Full: 16 Partial: 6 Not: 2 N/a: 2 |
* Statistical significance ( P < 0.05)
** Sum of scores on three-item questionnaire, max score, 12; lower values indicate higher health literacy.
↔: no change; ↑: higher value/score; ↓: lower value/score.
AS, aortic stenosis; CAD, coronary artery disease; CSE, cardiac self-efficacy; DAOH, days alive and out of hospital; DCS, decisional conflict scale; DM, diabetes mellitus; GAD-7, generalized anxiety disorder-7; IPDAS, International Patient Decision Aid Standards; NS, not significant ( P > 0.05); OMT, optimal medical therapy; PCI, percutaneous coronary intervention; PtDA, patient decision aid; RCT, randomized controlled trial; SD, standard deviation; SDM, shared decision-making; SAVR, surgical aortic valve replacement; TAVR, transcatheter aortic valve replacement; UC, usual care.
Studies reporting the development/acceptability of patient decision aids
One study 35 described the development of a PtDA for AS (TAVI vs. SAVR 34 ) that is no longer available, and two studies 54 , 59 described the development and acceptability of PtDAs for chronic CAD (PCI vs. medicines only; PCI Choice 58 , 61 and CONNECT 53 ). The systematic method of PtDA development recommended by IPDAS was implemented in the two CAD PtDA studies, 54 , 59 but only the CONNECT development study 54 cited a theory underpinning the methodology (i.e. Ottawa Decision Support Framework 65 ). Patients and/or healthcare professionals were involved in either providing feedback or user testing PtDAs across all development studies. 35 , 54 , 59 Methods included semi-structured interviews, 35 cognitive interviews, 54 video and teleconference calls, 35 , 54 focus groups, 54 , 59 and observations. 59 Participant demographics were only reported in the CONNECT PtDA study, which was the only study that assessed health literacy levels with 71% of participants scoring ‘adequate’ on the Brief Health Literacy Screen. 54
Studies evaluating the effectiveness of patient decision aids
Three AS PtDAs 39 , 41 , 43 and seven PtDAs for chronic CAD 50 , 55 , 58 , 61 , 62 , 64 were evaluated across seven studies. 20 , 40 , 42 , 51 , 56 , 60 , 63 Sample size ranged from 12 to 203 participants. Most participants were White and had an advanced level of education (i.e. completed college). A variety of decision-making processes and decisional quality outcomes were assessed, including, patient satisfaction, treatment preference, patient-centred communication, involvement in SDM, decisional conflict, and knowledge level. Two 20 , 56 out of four studies that measured the SDM process (via the OPTION Scale 66 or Control Preferences Scale 67 ) showed a significant improvement after using a PtDA for AS (TAVI or symptom management/palliative care 43 ) and CAD (PCI, medical therapy, or CABG 55 ). High scores for patient satisfaction, patient-centred communication (measured using CollaboRATE 68 ), and the Preparation for Decision-making Scale 69 were reported after PtDA use for both AS and chronic CAD treatments. 20 , 40 , 42 , 63 Patients’ treatment preference, treatment delivered, or treatment concordance with patient preferences did not significantly change in any study. 42 , 51 , 56 Cardiologists in two studies felt that they already performed SDM consistently and that PtDAs were poorly understood by patients and negatively impacted on consultations. 20 , 60 Most patients preferred a DVD- or booklet-formatted PtDAs than web-based formats. 56 , 63
Quality of studies
The 26-item SUNDAE checklist was used to evaluate the quality of reporting for all included studies, with results summarized in Table 3 (full evaluation in Supplementary material online , Table S12 ). Across the studies, between 50% and 89% (median 73%) of the SUNDAE criteria were completely fulfilled. Two of the three development studies either fully, or partially, satisfied all applicable SUNDAE. 54 , 59 No evaluation study achieved all 26 criteria. One criterion (Item 18) was only fully achieved by one study, 40 because the other six evaluation studies used a bespoke patient knowledge questionnaire, which had not undergone psychometric testing. Nine SUNDAE criteria were achieved by all studies. The criteria least consistently achieved were those related to the methods and results sections (e.g. ‘description of the development process’, ‘PtDA fidelity’, ‘process evaluation’, and ‘theories/models used to guide the study design and selection of evaluation measures’).
Meta-analyses
All six evaluation studies were assessed for inclusion in meta-analyses. Usable post-test data for patient knowledge and decisional conflict scores were obtained from four studies, with a total sample of 476 participants, 20 , 42 , 56 , 60 evaluating two PtDAs for AS 39 , 41 and three for chronic CAD. 55 , 58 , 61 Variation in the PtDAs and the patient groups across the four studies necessitated the use of standardized measures in the meta-analyses. Leave-one-out sensitivity analyses revealed no individual study to be exerting excessive influence on either meta-analysis (see Supplementary material online ).
Patient knowledge
Patient knowledge of treatment options was significantly greater in the PtDA groups compared with usual care in all four studies. 20 , 42 , 56 , 60 The meta-analysis determined that the synthesized estimate of the standardized mean difference in knowledge scores (PtDA—usual care) was 0.620 (95% CI 0.396–0.845), favouring the PtDA over usual care groups. A Z -test of the standardized mean effect indicated strong evidence at the 5% significance level for a non-zero effect ( Z = 5.42; P < 0.001). Cochran’s χ 2 test for heterogeneity indicated no evidence for statistical heterogeneity ( χ 2 (3) = 4.12; P = 0.248). The I 2 statistic was 27.3%, which may indicate low levels of heterogeneity. Data are summarized in Figure 2 .

Forest plot for the meta-analysis of patient knowledge scores.
Decisional conflict
Decisional conflict (measured by the validated SURE score 70 or Decisional Conflict Scale 71 ) was not significantly different between PtDA and usual care groups in all four studies. 20 , 42 , 56 , 60 However, the ‘informed’ subscale of the Decisional Conflict Scale score was significantly lower (i.e. favourable) in the PtDA groups compared with usual care. 56 , 60 The meta-analysis determined that the synthesized estimate of the standardized mean difference in decisional conflict (PtDA—usual care) was −0.159 (95% CI −0.339 to 0.022). A Z -test of the standardized mean effect revealed no evidence for a non-zero effect ( Z = −1.717; P = 0.086). Cochran’s χ 2 test for heterogeneity indicated no evidence for statistical heterogeneity (χ 2 (3) = 0.47; P = 0.925). The I 2 statistic was 0.00%, indicating that heterogeneity might not be important. Data are summarized in Figure 3 .

Forest plot for meta-analysis of decisional conflict scores.
Risk of bias
The RoB2 tool 32 was used to evaluate potential bias in the two randomized controlled studies 42 , 60 with results indicating ‘some concerns’ ( Figure 4 ). The two non-randomized studies 20 , 56 were evaluated using the NHLBI Quality Assessment of Controlled Intervention Studies and were rated as ‘fair quality’, indicating susceptibility to ‘some bias’. 33

Risk of bias summary using the Cochrane RoB2 tool.
Patient decision aids are evidence-based tools known to be effective in improving the quality of SDM to help patients receive care that is ‘right’ for them. Patients who use PtDAs are more knowledgeable, informed, and involved, have more accurate risk perceptions, and are more confident in their treatment decision and clearer about their health goals and treatment preferences. 18 This benefits patients because those who are more active in making treatment decisions tend to have better health outcomes and are more satisfied with their care. 72 Within cardiology, many patients with AS and chronic CAD have unresolved decisional needs and require support when considering treatment with TAVI and planned PCI, respectively. 14–17 Patient decision aids offer a potential solution but cardiology teams’ lack of awareness of available high-quality PtDAs is a barrier to implementation. 21
To the best of our knowledge, this review makes a useful contribution to the research literature as the first study to systematically identify and evaluate the availability, characteristics, and quality of PtDAs used to support SDM for AS and chronic CAD. We also report on the effectiveness of TAVI PtDAs to improve decisional quality, which extends an existing meta-analysis on SDM in cardiology settings that did not include this common interventional procedure. 22 These findings, combined with our narrative summary of PtDA evaluation and development studies, provide a comprehensive international overview of AS and CAD PtDAs to inform cardiology practice.
Patient decision aid availability and quality
Our findings on the availability of PtDAs ( Table 1 ) provide a valuable reference for cardiology teams and make an important contribution to the international literature. For the first time, internationally accepted quality criteria were used to evaluate the quality of AS and CAD PtDAs. We identified 21 PtDAs, but only one AS 41 and one CAD PtDA 49 were less than 5 years old and currently publicly available for patient distribution. However, only the AS PtDA was rated as high-quality having fulfilled all quality criteria. Given that SDM is recommended in clinical guidelines and health policy, 5 , 6 , 9 , 10 this lack of publicly available high-quality AS and CAD PtDAs is a significant finding that has not previously been reported. Overall, PtDAs scored poorly on criteria that address potentially harmful bias, which is consistent with reviews of cancer PtDAs. 23 This highlights that information concerning the uncertainty of treatment options, funding sources and updated policies, requires improvement. Doctors may be reluctant to discuss uncertainties around treatment outcomes, as they believe this will be viewed as incompetence 73 and will reduce patient trust and satisfaction with care. 74 Yet, from a patient perspective, higher levels of trust in cardiologists are associated with feeling listened to and involved in decisions about their health and treatments. 75 Having an open and honest dialogue is valued by heart disease patients. 76 Increasing cardiology teams’ awareness about patients’ communication preferences and additional SDM skills may improve this important element of SDM. 77
Patient decision aid accessibility
The PtDAs identified in this review had different designs, formats, and delivery approaches. There was a lack of consensus about the optimum characteristics for AS and CAD PtDAs. Potentially, this might be because patients’ and cardiology teams’ preferences varied; a view confirmed in this review. 54 , 56 , 63 A recent meta-analysis reported that the PtDA format (e.g. paper, computer, and web based) had no impact on effectiveness for improving SDM in cardiology settings. 22 Our results corroborate this finding; patient knowledge and some aspects of the SDM process (patient perception of SDM and integration of SDM in consultations) were significantly improved in two studies despite using PtDAs with different formats 20 , 56 : a printed one-page within-consultation ‘encounter PtDA’ for AS 43 and a web-based pre-consultation PtDA for CAD. 55 This suggests that a paper-based PtDA may be as effective as a more sophisticated digital version. However, additional research is required to corroborate this finding given the paucity of studies. We suggest that paper versions of PtDAs could be made routinely available, as a minimum, to support SDM for two reasons. First, 6–7% of adults in the USA 78 and the UK 79 have never used the internet. Second, it is recognized that the introduction of digital interventions can potentially widen health inequalities. 80
The overall quality of reporting, in both AS and CAD PtDA development and evaluation studies, was good, according to the recommended SUNDAE criteria. The aims, rationale, explanation of the PtDA and study methods, and implications for practice and research were comprehensively described in most studies. However, most studies did not measure PtDA fidelity or explore potential mechanisms for their effect on decision outcomes. The demographics of patients involved in the development and/or evaluation studies were either unknown, 35 , 59 under-reported, 40 , 51 or predominantly White, English-speaking people educated to high school level or higher. 20 , 42 , 54 , 56 , 60 , 63 Furthermore, readability levels were not reported in any PtDA, although the target reading age for two CAD PtDAs was reported as 13–14 years in associated publications. 54 , 56 These findings are significant because it is unclear how relevant and accessible existing AS and CAD PtDAs are for under-represented populations, which makes it challenging for cardiology teams to evaluate their appropriateness and usefulness within their clinical setting. Since patient–healthcare professional communication has the potential to reduce or increase health disparities, 81 it is important that the development and testing of PtDAs involve patients from diverse backgrounds.
Comparisons with other meta-analyses
Our meta-analyses found significantly improved levels of patient knowledge following the use of two AS PtDAs 39 , 41 and three CAD PtDAs, 55 , 58 , 61 compared with usual care. This finding is consistent with a recent meta-analysis of cardiology PtDAs. 22 However, our meta-analysis found no significant difference in decisional conflict between PtDA and usual care groups, in contrast to other reviews. 18 , 22 There are several potential explanations for this finding. The five PtDAs 41 , 43 , 55 , 58 , 61 evaluated may have limited function in eliciting preferences. Decisional conflict may have already been low in participants at baseline and/or in usual care groups 7 , 47 , 64 , 75 or the measure may have a ceiling effect. Another explanation relates to educational attainment. A large proportion of participants across the four studies had achieved a high-school education level or higher, which is known to be associated with lower decisional conflict. 82
Although not included in our meta-analysis due to heterogeneity of study designs, outcome measures indicating the quality of the decision-making process were significantly greater following the use of PtDAs across some 20 , 40 , 42 , 56 , 63 but not all studies, 51 , 60 and no negative outcomes were reported. The inconsistent findings might be explained by differences are study designs, outcomes, measurement instruments, and the PtDAs themselves. Given the wide variety of measures used to evaluate the quality of SDM, consensus on the most appropriate is recommended.

Implementation of patient decision aids in clinical practice
None of the PtDAs were evaluated in a large-scale randomized controlled trial that appeared to be sufficiently powered with a low risk of bias, possibly due to difficulties with recruitment and/or PtDA implementation. Several factors influence the successful implementation of PtDAs; a PtDA that is too complex or competes with existing practice is unlikely to be used. 77 Involvement and commitment from senior leadership and the clinical teams are an enabler to the use of PtDAs as is the engagement of the family and significant others. 77 Successful strategies to integrate PtDAs into clinical settings include training the entire cardiology team, linking PtDA outcomes with organizational priorities, proactively encouraging patients to engage with the PtDA, and reflecting on existing pathways to identify opportunities for PtDA use and SDM conversations. 77 The latter strategy could be particularly useful for elective PCI where the timing of PtDA delivery is challenging because diagnosis and treatment often occur together in the same procedure. 83 Providing PtDAs and seeking patients’ treatment preferences and goals earlier in the severe AS pathway should be considered. 13
Strengths and limitations
We comprehensively and systematically searched multiple databases, trial registers, and 30 online sources to identify AS and CAD PtDAs and their development and evaluation studies. However, we may not have identified all eligible PtDAs and six were not available so an evaluation of their characteristics and quality was not possible. The wide range of measurement instruments used to evaluate the quality of SDM limited the number of meta-analyses conducted and made cross study comparisons challenging. Nevertheless, this review provides a high-quality international review of AS and CAD PtDAs.
A diverse range of AS and CAD PtDAs has been developed over the past 16 years, but few are up to date and currently available. To increase the transparency around PtDA quality and effectiveness, information about the uncertainty of treatment outcomes, funding sources and future updates should be added. The ‘voice’ of underserved populations and those with low health literacy levels is needed in the development or evaluation of PtDAs as to date, this has been lacking. Paper-based versions of digital PtDAs should be available to avoid widening health inequalities associated with the digital divide. We recommend that cardiology teams use the most up-to-date and highest-quality PtDAs available. We concluded that patients who use PtDAs when considering treatments for AS or chronic CAD are likely to be better informed than those who do not.
Supplementary material is available at European Journal of Cardiovascular Nursing online.
We would like to thank Professor Richard Thomson for his expert guidance and support in the development of this review. We send thanks to Ellie Price for conducting the initial search of clinical trial registers. We would also like to thank the authors who provided further details about their PtDA and research study.
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
E.H., F.A., and D.C.: conceptualization; E.H. and F.A.: methodology; E.H. and J.S.: formal analysis; E.H., F.A., A.B., D.C., A.-Y.C., and H.C.: investigation; E.H.: project administration; E.H., F.A., and J.S.: visualization; E.H., F.A., and J.S.: writing—original draft; and E.H., F.A., A.B., J.S., D.C., A.-Y.C., and H.C.: writing—review & editing.
The data underlying this review are available in the article and in its online supplementary material.
iData Research . Interventional Cardiology Procedure Volume Analysis, Global, 2021–2027. In. BC, Canada; 2021. Available from https://idataresearch.com/product/interventional-cardiology-procedure-volume-analysis/ .
Patterson T , Allen CJ , Aroney N , Redwood S , Prendergast B . The future of transcatheter interventions . JACC Case Rep 2020 ; 2 : 2281 – 2282 .
Google Scholar
Leon MB , Smith CR , Mack M , Miller DC , Moses JW , Svensson LG , et al. Transcatheter aortic-valve implantation for aortic stenosis in patients who cannot undergo surgery . N Engl J Med 2010 ; 363 : 1597 – 1607 .
Spertus JA , Jones PG , Maron DJ , O’Brien SM , Reynolds HR , Rosenberg Y , et al. Health-Status outcomes with invasive or conservative care in coronary disease . N Engl J Med 2020 ; 382 : 1408 – 1419 .
Neumann F-J , Sousa-Uva M , Ahlsson A , Alfonso F , Banning AP , Benedetto U , et al. 2018 ESC/EACTS guidelines on myocardial revascularization . Eur Heart J 2019 ; 40 : 87 – 165 .
Vahanian A , Beyersdorf F , Praz F , Milojevic M , Baldus S , Bauersachs J , et al. 2021 ESC/EACTS guidelines for the management of valvular heart disease . Eur Heart J 2022 ; 43 : 561 – 632 .
Elwyn G , Frosch D , Rollnick S . Dual equipoise shared decision making: definitions for decision and behaviour support interventions . Implement Sci 2009 ; 4 : 75 .
National Institute for Health and Care Excellence (NICE) . Shared decision making: NICE guideline . In. UK : NICE ; 2021 .
Google Preview
Lawton JS , Tamis-Holland JE , Bangalore S , Bates ER , Beckie TM , Bischoff JM , et al. 2021 ACC/AHA/SCAI guideline for coronary artery revascularization . J Am Coll Cardiol 2022 ; 79 : e21 – e129 .
Otto CM , Nishimura RA , Bonow RO , Carabello BA , Erwin JP , Gentile F , et al. 2020 ACC/AHA guideline for the management of patients with valvular heart disease . J Am Coll Cardiol 2021 ; 77 : e25 – e197 .
Sepucha KR , Borkhoff CM , Lally J , Levin CA , Matlock DD , Ng CJ , et al. Establishing the effectiveness of patient decision aids: key constructs and measurement instruments . BMC Med Inform Decis Mak 2013 ; 13 : S12 .
Witteman HO , Ndjaboue R , Vaisson G , Dansokho SC , Arnold B , Bridges JFP , et al. Clarifying values: an updated and expanded systematic review and meta-analysis . Med Decis Making 2021 ; 41 : 801 – 820 .
van Beek-Peeters JJAM , van der Meer JBL , Faes MC , de Vos AJBM , van Geldorp MWA , Van den Branden BJL , et al. Professionals’ views on shared decision-making in severe aortic stenosis . Heart 2022 ; 108 : 558 – 564 .
Astin F , Stephenson J , Probyn J , Holt J , Marshall K , Conway D . Cardiologists’ and patients’ views about the informed consent process and their understanding of the anticipated treatment benefits of coronary angioplasty: a survey study . Eur J Cardiovasc Nurs 2020 ; 19 : 260 – 268 .
Probyn J , Greenhalgh J , Holt J , Conway D , Astin F . Percutaneous coronary intervention patients’ and cardiologists’ experiences of the informed consent process in Northern England: a qualitative study . BMJ Open 2017 ; 7 : e015127 .
Dharmarajan K , Foster J , Coylewright M , Green P , Vavalle JP , Faheem O , et al. The medically managed patient with severe symptomatic aortic stenosis in the TAVR era: patient characteristics, reasons for medical management, and quality of shared decision making at heart valve treatment centers . PLoS One 2017 ; 12 : e0175926 .
Col NF , Otero D , Lindman BR , Horne A , Levack MM , Ngo L , et al. What matters most to patients with severe aortic stenosis when choosing treatment? Framing the conversation for shared decision making . PLoS One 2022 ; 17 : e0270209 .
Stacey D , Legare F , Lewis K , Barry MJ , Bennett CL , Eden KB , et al. Decision aids for people facing health treatment or screening decisions . Cochrane Database Syst Rev 2017 ; 4 : CD001431 .
Stacey D , Suwalska V , Boland L , Lewis KB , Presseau J , Thomson R . Are patient decision aids used in clinical practice after rigorous evaluation? A survey of trial authors . Med Decis Making 2019 ; 39 : 805 – 815 .
Coylewright M , O'Neill E , Sherman A , Gerling M , Adam K , Xu K , et al. The learning curve for shared decision-making in symptomatic aortic stenosis . JAMA Cardiol 2020 ; 5 : 442 – 448 .
Nichols EL , Elwyn G , DiScipio A , Sidhu MS , O'Malley AJ , Matlock DD , et al. Cardiology providers’ recommendations for treatments and use of patient decision aids for multivessel coronary artery disease . BMC Cardiovasc Disord 2021 ; 21 : 410 .
Mitropoulou P , Grüner-Hegge N , Reinhold J , Papadopoulou C . Shared decision making in cardiology: a systematic review and meta-analysis . Heart 2022 ; 109 : 34 – 39 .
Grüne B , Kriegmair MC , Lenhart M , Michel MS , Huber J , Köther AK , et al. Decision aids for shared decision-making in uro-oncology: a systematic review . Eur Urol Focus 2022 ; 8 : 851 – 869 .
Torres Roldan VD , Brand-McCarthy SR , Ponce OJ , Belluzzo T , Urtecho M , Espinoza Suarez NR , et al. Shared decision making tools for people facing stroke prevention strategies in atrial fibrillation: a systematic review and environmental scan . Med Decis Making 2021 ; 41 : 540 – 549 .
Higgins JPT , Thomas J , Chandler J , Cumpston M , Li T , Page MJ , et al. Cochrane handbook for systematic reviews of interventions . Cochrane ; 2022 . version 6.4 . Available from www.training.cochrane.org/handbook .
Page MJ , McKenzie JE , Bossuyt PM , Boutron I , Hoffmann TC , Mulrow CD , et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews . BMJ 2021 ; 372 : n71 .
Elwyn G , O'Connor A , Stacey D , Volk R , Edwards A , Coulter A , et al. Developing a quality criteria framework for patient decision aids: online international Delphi consensus process . BMJ 2006 ; 333 : 417 .
DerSimonian R , Laird N . Meta-analysis in clinical trials . Control Clin Trials 1986 ; 7 : 177 – 188 .
StataCorp . Stata statistical software: release 17 . College Station, TX : StataCorp LLC ; 2021 .
Joseph-Williams N , Newcombe R , Politi M , Durand MA , Sivell S , Stacey D , et al. Toward Minimum standards for certifying patient decision aids: a modified delphi consensus process . Med Decis Making 2014 ; 34 : 699 – 710 .
Sepucha KR , Abhyankar P , Hoffman AS , Bekker HL , LeBlanc A , Levin CA , et al. Standards for UNiversal reporting of patient Decision Aid Evaluation studies: the development of SUNDAE checklist . BMJ Qual Saf 2018 ; 27 : 380 – 388 .
Sterne JAC , Savović J , Page MJ , Elbers RG , Blencowe NS , Boutron I , et al. Rob 2: a revised tool for assessing risk of bias in randomised trials . BMJ 2019 ; 366 : l4898 .
NHLBI Risk Assessment Work Group . Assessing cardiovascular risk systematic evidence review from the risk assessment work group . Bethesda, MD : National Heart, Lung, and Blood Institute ; 2013 .
Duke University . ADVICE: Navigating Aortic Valve Treatment Choices. https://www.pcori.org/research-results/2013/comparing-two-treatments-aortic-valve-disease
Brennan JM , Dokholyan RS , Graham F , Thomas L , Cohen DJ , Shahian D , et al. Comparing two treatments for aortic valve disease . Washington, DC, USA : Patient-Centered Outcomes Research Institute (PCORI): Duke University ; 2020 . doi: 10.25302/08.2020.CER-1306-04350
Lauck S , Borregaard B , Lewis K , De Souza I . Aortic Stenosis Choice (CHOICE-AS) .
Lauck S , Borregaard B , Lewis K , De Souza I . Implementation of shared decision-making for aortic stenosis: development of a patient decision aid . Eur J Cardiovasc Nurs 2021 ; 20 : zvab060.027 .
Col N . Aortic valve improved treatment approaches (AVITA) tool. https://classic.clinicaltrials.gov/ct2/show/NCT04755426
American College of Cardiology . A decision aid for treatment options for severe aortic stenosis (TAVI vs Symptom Management). https://www.cardiosmart.org/assets/decision-aid/choosing-between-tavr-and-symptom-management
Einfeld K . Implementation of peer support and shared decision-making aids for the transcatheter aortic valve replacement population . Boise, Idaho, USA : Boise State University, School of Nursing ; 2020 .
American College of Cardiology . A decision aid for treatment options for severe aortic stenosis for patients deciding between TAVI and surgery. https://www.cardiosmart.org/assets/decision-aid/choosing-between-tavr-and-surgery
Valentine KD , Marques F , Selberg A , Flannery L , Langer N , Inglessis I , et al. Impact of decision aid on decision-making of patients with severe aortic stenosis: randomized pilot study . JSCAI 2022 ; 1 : 100025 .
American College of Cardiology . Severe Aortic Stenosis Decision Aid. https://sharedcardiology.org/tools/
MAGIC Evidence Ecosystem Foundation (BMJ RapidRecs) . TAVI versus SAVR for patients with severe symptomatic aortic stenosis at low to intermediate perioperative risk: for patients above 85 years with severe symptomatic aortic stenosis, at low or intermediate perioperative risk. https://app.magicapp.org/#/guideline/1308
MAGIC Evidence Ecosystem Foundation (BMJ RapidRecs) . TAVI versus SAVR for patients with severe symptomatic aortic stenosis at low to intermediate perioperative risk: for patients 75–85 years with severe symptomatic aortic stenosis who are at low or intermediate perioperative risk. https://app.magicapp.org/#/guideline/1308
MAGIC Evidence Ecosystem Foundation (BMJ RapidRecs) . TAVI versus SAVR for patients with severe symptomatic aortic stenosis at low to intermediate perioperative risk: for patients aged 65 to < 75 years and eligible for transfemoral TAVI or SAVR. https://app.magicapp.org/#/guideline/1308
MAGIC Evidence Ecosystem Foundation (BMJ RapidRecs) . TAVI versus SAVR for patients with severe symptomatic aortic stenosis at low to intermediate perioperative risk: for patients aged < 65 years and eligible for transfemoral TAVI or SAVR. https://app.magicapp.org/#/guideline/1308
MAGIC Evidence Ecosystem Foundation (BMJ RapidRecs) . TAVI versus SAVR for patients with severe symptomatic aortic stenosis at low to intermediate perioperative risk who cannot undergo transfemoral TAVR but can undergo transapical approach. https://app.magicapp.org/#/guideline/1308
Mayo Clinic . Angina treatment: Stents, drugs, lifestyle changes—What's best? https://www.mayoclinic.org/diseases-conditions/coronary-artery-disease/in-depth/angina-treatment/art-20046240
Option Grid Collaborative . Angina: treatment options Option Grid™ .
Scalia P , Durand MA , Kremer J , Faber M , Elwyn G . Online, interactive option grid patient decision aids and their effect on user preferences . Med Decis Making 2018 ; 38 : 56 – 68 .
DynaMed Decisions EBSCO Health . Chest Pain (Stable Angina) Treatment Options Option Grid™. www.ebsco.com
Harris E , Conway D , Jimenez-Aranda A , Butts J , Hedley-Takhar P , Thomson R , et al. CONNECT: COroNary aNgioplasty dECision Tool .
Harris E , Conway D , Jimenez-Aranda A , Butts J , Hedley-Takhar P , Thomson R , et al. Development and user-testing of a digital patient decision aid to facilitate shared decision-making for people with stable angina . BMC Med Inform Decis Mak 2022 ; 22 : 143 .
Duke University Medical Center clinicians and Healthwise . Coronary Artery Disease: What treatment would you prefer?
Doll JA , Jones WS , Lokhnygina Y , Culpepper S , Parks RL , Calhoun C , et al. PREPARED study: a study of shared decision-making for coronary artery disease . Circ Cardiovasc Qual Outcomes 2019 ; 12 : e005244 .
NHS England Vale of York Clinical Commissioning Group . Deciding what to do about stable angina. https://www.valeofyorkccg.nhs.uk/rss/home/patient-decision-making/shared-decision-making/
Mayo Foundation for Medical Education and Research . PCI Choice: Class I/II Stable Angina. https://carethatfits.org/pci-choice/
Coylewright M , Shepel K , Leblanc A , Pencille L , Hess E , Shah N , et al. Shared decision making in patients with stable coronary artery disease: PCI choice . PLoS One 2012 ; 7 : e49827 .
Coylewright M , Dick S , Zmolek B , Askelin J , Hawkins E , Branda M , et al. PCI choice decision aid for stable coronary artery disease: a randomized trial . Circ Cardiovasc Qual Outcomes 2016 ; 9 : 767 – 776 .
Mayo Foundation for Medical Education and Research . PCI Choice: Class III Stable Angina. 2012 . https://carethatfits.org/pci-choice/
Healthwise . Should I Have Angioplasty for Stable Chest Angina? https://decisionaid.ohri.ca/Azsumm.php? ID=1202
Hinsberg L , Marques F , Leavitt L , Skubisz C , Sepucha K , Wasfy JH . Comparing the effectiveness of two different decision aids for stable chest discomfort . Coron artery dis 2018 ; 29 : 230 – 236 .
Health Dialog and Foundation for Informed Medical Decision Making. Treatment Choices for Stable Chest Discomfort .
Stacey D , Légaré F , Boland L , Lewis KB , Loiselle M-C , Hoefel L , et al. 20th anniversary Ottawa decision support framework: part 3 overview of systematic reviews and updated framework . Med Decis Making 2020 ; 40 : 379 – 398 .
Elwyn G , Hutchings H , Edwards A , Rapport F , Wensing M , Cheung WY , et al. The OPTION scale: measuring the extent that clinicians involve patients in decision-making tasks . Health Expect 2005 ; 8 : 34 – 42 .
Degner LF , Sloan JA , Venkatesh P . The control preferences scale . Can J Nurs Res 1997 ; 29 : 21 – 43 .
Elwyn G , Barr PJ , Grande SW , Thompson R , Walsh T , Ozanne EM . Developing CollaboRATE: a fast and frugal patient-reported measure of shared decision making in clinical encounters . Patient Educ Couns 2013 ; 93 : 102 – 107 .
Bennett C , Graham ID , Kristjansson E , Kearing SA , Clay KF , O'Connor AM . Validation of a preparation for decision making scale . Patient Educ Couns 2010 ; 78 : 130 – 133 .
Legare F , Kearing S , Clay K , Gagnon S , D'Amours D , Rousseau M , et al. Are you SURE?: assessing patient decisional conflict with a 4-item screening test . Can Fam Physician 2010 ; 56 : e308 – e314 .
O'Connor AM . Validation of a decisional conflict scale . Med Decis Making 1995 ; 15 : 25 – 30 .
Hibbard JH , Greene J . What the evidence shows about patient activation: better health outcomes and care experiences; fewer data on costs . Health Aff (Millwood) 2013 ; 32 : 207 – 214 .
Zeuner R , Frosch DL , Kuzemchak MD , Politi MC . Physicians’ perceptions of shared decision-making behaviours: a qualitative study demonstrating the continued chasm between aspirations and clinical practice . Health Expect 2015 ; 18 : 2465 – 2476 .
Henry MS . Uncertainty, responsibility, and the evolution of the physician/patient relationship . J Med Ethics 2006 ; 32 : 321 – 323 .
Keating NL , Gandhi TK , Orav EJ , Bates DW , Ayanian JZ . Patient characteristics and experiences associated with trust in specialist physicians . Arch Intern Med 2004 ; 164 : 1015 – 1020 .
Mentrup S , Harris E , Gomersall T , Köpke S , Astin F . Patients’ experiences of cardiovascular health education and risk communication: a qualitative synthesis . Qual Health Res 2020 ; 30 : 88 – 104 .
Joseph-Williams N , Abhyankar P , Boland L , Bravo P , Brenner AT , Brodney S , et al. What works in implementing patient decision aids in routine clinical settings? A rapid realist review and update from the International Patient Decision Aid Standards Collaboration . Med Decis Making 2021 ; 41 : 907 – 937 .
Pew Research Center . Survey of U.S. adults conducted Jan. 25-Feb, 8, 2021 . Washington, DC, USA : Pew Research Center ; 2021 .
Office for National Statistics . Internet users 2020 . Newport, South Wales, UK : Office for National Statistics ; 2021 . https://www.ons.gov.uk/businessindustryandtrade/itandinternetindustry/datasets/internetusers .
Yao R , Zhang W , Evans R , Cao G , Rui T , Shen L . Inequities in health care services caused by the adoption of digital health technologies: scoping review . J Med Internet Res 2022 ; 24 : e34144 .
Pérez-Stable EJ , El-Toukhy S . Communicating with diverse patients: how patient and clinician factors affect disparities . Patient Educ Couns 2018 ; 101 : 2186 – 2194 .
Tang H , Wang S , Dong S , Du R , Yang X , Cui P , et al. Surgery decision conflict and its related factors among newly diagnosed early breast cancer patients in China: a cross-sectional study . Nurs Open 2021 ; 8 : 2578 – 2586 .
Peters LJ , Torres-Castaño A , van Etten-Jamaludin FS , Perestelo Perez L , Ubbink DT . What helps the successful implementation of digital decision aids supporting shared decision-making in cardiovascular diseases? A systematic review . Eur Heart J Digit Health 2023 ; 4 : 53 – 62 .
Author notes
- aortic valve stenosis
- coronary arteriosclerosis
- decision support techniques
Supplementary data
| Month: | Total Views: |
|---|---|
| December 2023 | 43 |
| January 2024 | 172 |
| February 2024 | 103 |
| March 2024 | 130 |
| April 2024 | 101 |
| May 2024 | 106 |
| June 2024 | 95 |
| July 2024 | 117 |
| August 2024 | 106 |
| September 2024 | 44 |
Email alerts
Companion article.
- Power to the people? Time to improve and implement patient decision aids to strengthen shared decision-making
Related articles in pubmed, citing articles via.
- Recommend to Your Librarian
- Advertising and Corporate Services
- Journals Career Network
Affiliations
- Online ISSN 1873-1953
- Print ISSN 1474-5151
- Copyright © 2024 European Society of Cardiology
- About Oxford Academic
- Publish journals with us
- University press partners
- What we publish
- New features
- Open access
- Institutional account management
- Rights and permissions
- Get help with access
- Accessibility
- Advertising
- Media enquiries
- Oxford University Press
- Oxford Languages
- University of Oxford
Oxford University Press is a department of the University of Oxford. It furthers the University's objective of excellence in research, scholarship, and education by publishing worldwide
- Copyright © 2024 Oxford University Press
- Cookie settings
- Cookie policy
- Privacy policy
- Legal notice
This Feature Is Available To Subscribers Only
Sign In or Create an Account
This PDF is available to Subscribers Only
For full access to this pdf, sign in to an existing account, or purchase an annual subscription.
Comparative Effectiveness of Decision Aids for Cancer-Screening Decision Making: An Overview of Reviews
- Systematic Review
- Published: 04 September 2024
Cite this article

- Masaya Hibino MD 1 ,
- Chisato Hamashima MD, PhD 2 ,
- Miyuki Hirosue MS 1 ,
- Mitsunaga Iwata MD, PhD 1 &
- Teruhiko Terasawa MD, PhD ORCID: orcid.org/0000-0002-0975-391X 1 , 3
1 Altmetric
Decision aids (DAs), compared to no DAs, help improve the key aspects of shared decision-making, including increased knowledge, discussion frequency, and reduction in decisional conflict. However, systematic reviews have reported varied conclusions on screening uptake, and which DAs are superior to alternative forms in shared decision-making for cancer screening has not been comprehensively reviewed.
An overview of systematic reviews was performed. Multiple databases were searched up to December 31, 2023, for systematic reviews of randomized controlled trials (RCTs) and non-randomized comparative studies (NRCSs) of any size that assessed a decision aid aimed to facilitate cancer-screening decision making communications. Dual screening of abstracts and full-text reports, dual data extraction and quality assessment, and qualitative synthesis were performed.
The 22 eligible publications included 24 reviews on cancer screening DAs for a single specific cancer (8, 8, 7, and 1 on prostate, breast, colorectal, and lung cancer, respectively) and three reviews on multiple aggregate cancers. Individual reviews were based on different primary study designs (92 RCTs and 37 NRCSs); each study was infrequently cited (median citation count 2; range 1–9). Although the DAs had variable formats and delivery methods, the reviews generally focused on use and non-use comparisons. DAs decreased the intention or actual uptake for prostate and breast cancer screening, but increased it for colorectal cancer screening. DAs were associated with increased knowledge, well-informed choice, and reduced decisional conflict, regardless of cancer type. Only four reviews on comparative effectiveness between alternative formats of DAs (based on 14 RCTs and 2 NRCSs) failed to conclude on the specific format that was superior to others.
DAs improve cancer screening shared decision-making by boosting cancer screening knowledge and informed choice and lowering decisional conflict and may facilitate preference-based, individualized screening participation. Comparative data on different cancer screening DAs are limited.
Systematic review registration
PROSPERO, CRD42021235957.
This is a preview of subscription content, log in via an institution to check access.
Access this article
Subscribe and save.
- Get 10 units per month
- Download Article/Chapter or eBook
- 1 Unit = 1 Article or 1 Chapter
- Cancel anytime
Price includes VAT (Russian Federation)
Instant access to the full article PDF.
Rent this article via DeepDyve
Institutional subscriptions
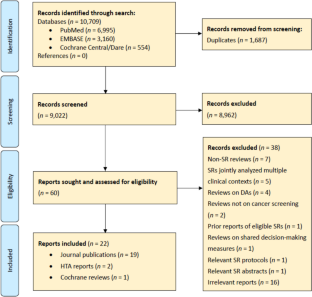
Similar content being viewed by others

The effects of shared decision-making compared to usual care for prostate cancer screening decisions: a systematic review and meta-analysis
Wilfully out of sight a literature review on the effectiveness of cancer-related decision aids and implementation strategies.

Shared medical decision in prostate cancer screening in primary care: a systematic literature review of current evidence
Data availability.
All data generated or analyzed during this study are included in this published article and its supplementary information files.
Marcus PM. Assessment of Cancer Screening: A Primer. Springer Nature, Cham; 2022. https://doi.org/10.1007/978-3-030-94577-0 .
Sheridan SL, Harris RP, Woolf SH. Shared decision making about screening and chemoprevention. a suggested approach from the U.S. Preventive Services Task Force. Am J Prev Med. 2004;26:56–6.
Stacey D, Légaré F, Lewis K, et al. Decision aids for people facing health treatment or screening decisions. Cochrane Database Syst Rev. 2017;4:Cd001431.
Wolf AM, Nasser JF, Wolf AM, Schorling JB. The impact of informed consent on patient interest in prostate-specific antigen screening. Arch Intern Med. 1996;156:1333–6.
Article CAS PubMed Google Scholar
Whitman A, De Lew N, Chappel A, Aysola V, Zuckerman R, Sommers BD. Addressing social determinants of health: Examples of successful evidence-based strategies and current federal efforts. Off Heal Policy. 2022;1:1-30.
Google Scholar
Ankolekar A, Dekker A, Fijten R, Berlanga A. The Benefits and Challenges of Using Patient Decision Aids to Support Shared Decision Making in Health Care. JCO Clin Cancer Inform. 2018;2:1-10.
Article PubMed Google Scholar
Muscat DM, Smith J, Mac O, et al. Addressing Health Literacy in Patient Decision Aids: An Update from the International Patient Decision Aid Standards. Med Decis Making. 2021;41(7):848-69.
Article PubMed PubMed Central Google Scholar
Cox CE, White DB, Abernethy AP. A universal decision support system. Addressing the decision-making needs of patients, families, and clinicians in the setting of critical illness. Am J Respir Crit Care Med. 2014;190(4):366–73.
Syrowatka A, Krömker D, Meguerditchian AN, Tamblyn R. Features of Computer-Based Decision Aids: Systematic Review, Thematic Synthesis, and Meta-Analyses. J Med Internet Res. 2016;18(1):e20.
Health Care Education Association. Patient Education Practice Guidelines for Health Care Professionals2021. https://www.hcea-info.org/assets/hcea%20guidelines_BW%201-25-2021.pdf Accessed 9 June 2024.
Gates M, Gates A, Pieper D, et al. Reporting guideline for overviews of reviews of healthcare interventions: development of the PRIOR statement. BMJ. 2022;378:e070849.
Hibino M, Hamashima C, Iwata M, Terasawa T. Effectiveness of decision aids on cancer-screening decision-making: an umbrella review protocol. BMJ Open. 2021;11:e051156.
Sterne JA, Hernán MA, Reeves BC, et al. ROBINS-I: a tool for assessing risk of bias in non-randomized studies of interventions. BMJ. 2016;355:i4919.
Ebell MH, Thai TN, Royalty KJ. Cancer screening recommendations: an international comparison of high income countries. Public Health Rev. 2018;39:7.
Wallace BC, Small K, Brodley CE, Lau J, Trikalinos TA. Deploying an interactive machine learning system in an evidence-based practice center: abstrackr. Proceedings of the 2nd ACM SIGHIT International Health Informatics Symposium. 2012;2012;819–24.
Shea BJ, Reeves BC, Wells G, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomized or non-randomized studies of healthcare interventions, or both. BMJ. 2017;358:j4008.
Guyatt GH, Oxman AD, Vist GE, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336:924–6.
Owens DK, Lohr KN, Atkins D, et al. AHRQ series paper 5: grading the strength of a body of evidence when comparing medical interventions--agency for healthcare research and quality and the effective healthcare program. J Clin Epidemiol. 2010;63:513–23.
Guyatt G, Oxman AD, Sultan S, et al. GRADE guidelines: 11. Making an overall rating of confidence in effect estimates for a single outcome and for all outcomes. J Clin Epidemiol. 2013;66:151–7.
Evans R, Edwards A, Brett J, et al. Reduction in uptake of PSA tests following decision aids: systematic review of current aids and their evaluations. Patient Educ Couns. 2005;58:13–26.
Volk RJ, Hawley ST, Kneuper S, et al. Trials of decision aids for prostate cancer screening: a systematic review. Am J Prev Med. 2007;33:428–34.
Ilic D, Jammal W, Chiarelli P, et al. Assessing the effectiveness of decision aids for decision making in prostate cancer testing: a systematic review. Psychooncology. 2015;24:1303–15.
Baptista S, Teles Sampaio E, Heleno B, Azevedo LF, Martins C. Web-based versus usual care and other formats of decision aids to support prostate cancer screening decisions: systematic review and meta-analysis. J Med Internet Res. 2018;20:e228.
Ivlev I, Jerabkova S, Mishra M, Cook LA, Eden KB. Prostate cancer screening patient decision aids: a systematic review and meta-analysis. Am J Prev Med. 2018;55:896–907.
Riikonen JM, Guyatt GH, Kilpeläinen TP, et al. Decision aids for prostate cancer screening choice: a systematic review and meta-analysis. JAMA Intern Med. 2019;179:1072–82.
Ivlev I, Hickman EN, McDonagh MS, Eden KB. Use of patient decision aids increased younger women’s reluctance to begin screening mammography: a systematic review and meta-analysis. J Gen Intern Med. 2017;32:803–12.
Martínez-Alonso M, Carles-Lavila M, Pérez-Lacasta MJ, Pons-Rodríguez A, Garcia M, Rué M. Assessment of the effects of decision aids about breast cancer screening: a systematic review and meta-analysis. BMJ Open. 2017; 7 :e016894.
Gao JP, Jin YH, Yu SF, Wu WF, Han SF. Evaluate the effectiveness of breast cancer decision aids: A systematic review and meta-analysis of randomize clinical trials. Nurs Open. 2021;8:2091–104.
Yu L, Li P, Yang S, et al. Web-based decision aids to support breast cancer screening decisions: systematic review and meta-analysis. J Comp Eff Res. 2020;9:985–1002.
Yu L, Yang S, Zhang C, et al. Decision aids for breast cancer screening in women approximately 50 years of age: a systematic review and meta-analysis of randomized controlled trials. J Clin Nurs. 2021. https://doi.org/10.1111/jocn.16112 . Online ahead of print.
Volk RJ, Linder SK, Lopez-Olivo MA, et al. Patient decision aids for colorectal cancer screening: a systematic review and meta-analysis. Am J Prev Med. 2016;51:779–91.
Ramli NS, Manaf MRA, Hassan MR, Ismail MI, Nawi AM. Effectiveness of colorectal cancer screening promotion using e-media decision aids: a systematic review and meta-analysis. Int J Environ Res Public Health. 2021;18:8190.
Larsen MB, Stokholm R, Kirkegaard P, Laursen HS, Gabel P, Andersen B. Making decisions on your own: self-administered decision aids about colorectal cancer screening - a systematic review and meta-analyses. Patient Educ Couns. 2022;105:534–46.
Fukunaga MI, Halligan K, Kodela J, et al. Tools to promote shared decision-making in lung cancer screening using low-dose CT scanning: a systematic review. Chest. 2020; 158 :2646–57.
O’Brien MA, Whelan TJ, Villasis-Keever M, et al. Are cancer-related decision aids effective? A systematic review and meta-analysis. J Clin Oncol. 2009; 27 :974–85.
van Agt HME, Korfage IJ, Essink-Bot M-L. Interventions to enhance informed choices among invitees of screening programmes—a systematic review. Eur J Public Health. 2014;24:789–801.
McAlpine K, Lewis KB, Trevena LJ, Stacey D. What Is the Effectiveness of Patient Decision Aids for Cancer-Related Decisions? A Systematic Review Subanalysis. JCO Clin Cancer Inform 2018;2:1-13.
Herrera DJ, van de Veerdonk W, Berhe NM, et al. Mixed-Method Systematic Review and Meta-Analysis of Shared Decision-Making Tools for Cancer Screening. Cancers (Basel). 2023;15(15):3867.
Lillie SE, Partin MR, Rice K, et al. The Effects of Shared Decision Making on Cancer Screening – A Systematic Review [Internet]. Washington (DC): Department of Veterans Affairs (US);2014.
Holden DJ, Harris R, Porterfield DS, et al. Enhancing the use and quality of colorectal cancer screening. Evidence Report/Technology Assessment No.190. AHRQ Publication, Rockville; 2010. https://www.ncbi.nlm.nih.gov/books/NBK44526/ .
Higgins JPT, Altman DG, Gøtzsche PC, et al. The Cochrane collaboration’s tool for assessing risk of bias in randomized trials. BMJ. 2011;343:d5928.
Taylor KL, Davis JL 3rd, Turner RO, et al. Educating African American men about the prostate cancer screening dilemma: a randomized intervention. Cancer Epidemiol Biomarkers Prev. 2006;15:2179–88.
Gattellari M, Ward JE. A community-based randomized controlled trial of three different educational resources for men about prostate cancer screening. Patient Educ Couns. 2005;57:168–82.
Frosch DL, Kaplan RM, Felitti VJ. A randomized controlled trial comparing internet and video to facilitate patient education for men considering the prostate specific antigen test. J Gen Intern Med. 2003;18:781–7.
Evans R, Joseph-Williams N, Edwards A, et al. Supporting informed decision making for prostate specific antigen (PSA) testing on the web: an online randomized controlled trial. J Med Internet Res. 2010;12:e27.
Krist AH, Woolf SH, Johnson RE, Kerns JW. Patient education on prostate cancer screening and involvement in decision making. Ann Fam Med. 2007; 5 :112–9.
Taylor KL, Williams RM, Davis K, et al. Decision making in prostate cancer screening using decision aids vs usual care: a randomized clinical trial. JAMA Intern Med. 2013;173:1704–12.
PubMed PubMed Central Google Scholar
Ilic D, Egberts K, McKenzie JE, Risbridger G, Green S. Informing men about prostate cancer screening: a randomized controlled trial of patient education materials. J Gen Intern Med. 2008;23:466–71.
Partin MR, Nelson D, Radosevich D, et al. Randomized trial examining the effect of two prostate cancer screening educational interventions on patient knowledge, preferences, and behaviors. J Gen Intern Med. 2004;19:835–42.
Partin MR, Nelson D, Flood AB, Friedemann-Sánchez G, Wilt TJ. Who uses decision aids? Subgroup analyses from a randomized controlled effectiveness trial of two prostate cancer screening decision support interventions. Health Expect. 2006; 9 :285–95.
Schroy PC 3rd, Emmons K, Peters E, et al. The impact of a novel computer-based decision aid on shared decision making for colorectal cancer screening: a randomized trial. Med Decis Making. 2011;31:93–107.
Jerant A, Kravitz RL, Rooney M, Amerson S, Kreuter M, Franks P. Effects of a tailored interactive multimedia computer program on determinants of colorectal cancer screening: a randomized controlled pilot study in physician offices. Patient Educ Couns 2007;66:67–74.
Schroy PC 3rd, Emmons KM, Peters E, et al. Aid-assisted decision making and colorectal cancer screening: a randomized controlled trial. Am J Prev Med. 2012;43:573–83.
Griffith JM, Fichter M, Fowler FJ, Lewis C, Pignone MP. Should a colon cancer screening decision aid include the option of no testing? A comparative trial of two decision aids. BMC Med Inform Decis Mak. 2008;8:10.
Griffith JM, Lewis CL, Brenner AR, Pignone MP. The effect of offering different numbers of colorectal cancer screening test options in a decision aid: a pilot randomized trial. BMC Med Inform Decis Mak. 2008; 8 :4.
Ruparel M, Quaife SL, Ghimire B, et al. Impact of a lung cancer screening information film on informed decision-making: a randomized trial. Ann Am Thorac Soc. 2019;16:744–51.
Tanner NT, Banas E, Yeager D, Dai L, Hughes Halbert C, Silvestri GA. In-person and telephonic shared decision-making visits for people considering lung cancer screening: an assessment of decision quality. Chest. 2019;155:236–8.
Shah SC, Kayamba V, Peek RM Jr, Heimburger D. Cancer control in low- and middle-income countries: is it time to consider screening? J Glob Oncol. 2019;5:1–8.
CAS PubMed Google Scholar
Welton NJ, Caldwell DM, Adamopoulos E, Vedhara K. Mixed treatment comparison meta-analysis of complex interventions: psychological interventions in coronary heart disease. Am J Epidemiol. 2009;169:1158–65.
Guise JM, Chang C, Viswanathan M, et al. AHRQ Methods for Effective Health Care. Systematic Reviews of Complex Multicomponent Health Care Interventions [Internet]. Rockville (MD): Agency for Healthcare Research and Quality (US);2014.
Petropoulou M, Efthimiou O, Rücker G, et al. A review of methods for addressing components of interventions in meta-analysis. PLoS One. 2021;16:e0246631.
Article CAS PubMed PubMed Central Google Scholar
Download references
Acknowledgements
The English language editing was provided by Editage ( http://www.editage.com ). This assistance was funded by the Sciences Research Grants for Promotion of Cancer Control Programs by the Ministry of Health, Labor and Welfare, Japan (grant number 20EA1024).
This research was supported by the Sciences Research Grants for Promotion of Cancer Control Programs by the Ministry of Health, Labor and Welfare, Japan (grant number 20EA1024).
Author information
Authors and affiliations.
Department of Emergency Medicine and General Internal Medicine, Fujita Health University School of Medicine, Toyoake, Aichi, Japan
Masaya Hibino MD, Miyuki Hirosue MS, Mitsunaga Iwata MD, PhD & Teruhiko Terasawa MD, PhD
Health Policy Section, Division of Nursing, Faculty of Medical Technology, Teikyo University, Tokyo, Japan
Chisato Hamashima MD, PhD
Section of General Internal Medicine, Department of Emergency Medicine and General Internal Medicine, Fujita Health University School of Medicine, Toyoake, Aichi, Japan
Teruhiko Terasawa MD, PhD
You can also search for this author in PubMed Google Scholar
Contributions
All the authors cited in the manuscript made substantial contributions to the concept and design, the execution of the work, or the analysis and interpretation of data; and to drafting or revising the manuscript; and have read and approved the final version of the paper. All authors had access to the data. Masaya Hibino and Teruhiko Terasawa verified the dataset. All authors were responsible for making the decision to submit this manuscript.
Masaya Hibino: conceptualization, data curation, formal analysis, investigation, validation, and writing — review and editing.
Chisato Hamashima: conceptualization, data curation, formal analysis, funding acquisition, investigation, methodology, project administration, resources, supervision, validation, and writing — review and editing.
Miyuki Hirosue: conceptualization, data curation, formal analysis, investigation, and writing — review and editing.
Mitsunaga Iwata: conceptualization, formal analysis, supervision, and writing — review and editing.
Teruhiko Terasawa: conceptualization, data curation, formal analysis, investigation, methodology, project administration, resources, software, supervision, validation, visualization, writing — original draft, and writing — review and editing.
Corresponding author
Correspondence to Teruhiko Terasawa MD, PhD .
Ethics declarations
Conflict of interest.
The authors declare that they do not have a conflict of interest.
The study funders had no role in study design, data collection, data analysis, data interpretation, writing of the manuscript, or the decision to submit the manuscript for publication.
Additional information
Publisher's note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This work was presented in part at the 82nd Annual Meeting of the Japanese Society of Public Health, held on October 31–November 2, 2023, in Tsukuba International Congress Center, Tsukuba, Japan.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (DOCX 2201 KB)
Supplementary file2 (docx 25 kb), rights and permissions.
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Reprints and permissions
About this article
Hibino, M., Hamashima, C., Hirosue, M. et al. Comparative Effectiveness of Decision Aids for Cancer-Screening Decision Making: An Overview of Reviews. J GEN INTERN MED (2024). https://doi.org/10.1007/s11606-024-09001-4
Download citation
Received : 13 March 2024
Accepted : 09 August 2024
Published : 04 September 2024
DOI : https://doi.org/10.1007/s11606-024-09001-4
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- cancer screening
- comparative effectiveness
- decision aids
- shared decision making
- systematic review
- Find a journal
- Publish with us
- Track your research
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
- My Bibliography
- Collections
- Citation manager
Save citation to file
Email citation, add to collections.
- Create a new collection
- Add to an existing collection
Add to My Bibliography
Your saved search, create a file for external citation management software, your rss feed.
- Search in PubMed
- Search in NLM Catalog
- Add to Search
End-of-life decision making by family caregivers of persons with advanced dementia: A literature review of decision aids
Affiliations.
- 1 School of Nursing, The University of Texas at Austin, Austin, TX, USA.
- 2 School of Information, The University of Texas at Austin, Austin, TX, USA.
- PMID: 29844911
- PMCID: PMC5966844
- DOI: 10.1177/2050312118777517
Objectives: To investigate existing knowledge in the literature about end-of-life decision making by family caregivers of persons with dementia, focusing on decision aids for caregivers of persons with advanced dementia, and to identify gaps in the literature that can guide future research.
Methods: A literature review through systematic searches in PubMed, CINAHL Plus with Full Text, and PsycINFO was conducted in February 2018; publications with full text in English and published in the past 10 years were selected in multiple steps.
Results: The final sample included five decision aids with predominantly Caucasian participants; three of them had control groups, and three used audiovisual technology in presenting the intervention materials. No other technology was used in any intervention. Existing interventions lacked tailoring of information to caregivers' preferences for different types and amounts of information necessary to make decisions consistent with patients' values.
Conclusion: Research is needed in exploring the use of technology in decision aids that could provide tailored information to facilitate caregivers' decision making. More diverse samples are needed.
Keywords: Alzheimer’s; Dementia; decision aids; end-of-life; family caregivers.
PubMed Disclaimer
Conflict of interest statement
Declaration of conflicting interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Systematic selection process.
Similar articles
- Decision Aid Interventions for Family Caregivers of Persons With Advanced Dementia in Decision-Making About Feeding Options: A Scoping Review. Pei Y, Qi X, Schulman-Green D, Hu M, Wang K, Wu B. Pei Y, et al. J Am Med Dir Assoc. 2022 Dec;23(12):1927.e1-1927.e6. doi: 10.1016/j.jamda.2022.08.014. Epub 2022 Sep 21. J Am Med Dir Assoc. 2022. PMID: 36150408 Free PMC article. Review.
- Screening for Cognitive Impairment in Older Adults: An Evidence Update for the U.S. Preventive Services Task Force [Internet]. Patnode CD, Perdue LA, Rossom RC, Rushkin MC, Redmond N, Thomas RG, Lin JS. Patnode CD, et al. Rockville (MD): Agency for Healthcare Research and Quality (US); 2020 Feb. Report No.: 19-05257-EF-1. Rockville (MD): Agency for Healthcare Research and Quality (US); 2020 Feb. Report No.: 19-05257-EF-1. PMID: 32129963 Free Books & Documents. Review.
- Older Persons' and Their Caregivers' Perspectives and Experiences of Research Participation With Impaired Decision-Making Capacity: A Scoping Review. Hosie A, Kochovska S, Ries N, Gilmore I, Parker D, Sinclair C, Sheehan C, Collier A, Caplan GA, Visser M, Xu X, Lobb E, Sheahan L, Brown L, Lee W, Sanderson CR, Amgarth-Duff I, Green A, Edwards L, Agar MR. Hosie A, et al. Gerontologist. 2022 Feb 9;62(2):e112-e122. doi: 10.1093/geront/gnaa118. Gerontologist. 2022. PMID: 32866239 Review.
- [The Impact of Family-Centered Advance Care Planning for Persons With Dementia and Family Caregivers on Decision-Making Conflicts in End-of-Life Care]. Lu WR, Liu CL, Zeng YF, Chang HJ, Liao W, Huang HL. Lu WR, et al. Hu Li Za Zhi. 2021 Feb;68(1):30-42. doi: 10.6224/JN.202102_68(1).06. Hu Li Za Zhi. 2021. PMID: 33521917 Chinese.
- Decision aids to support decision-making in dementia care: a systematic review. Davies N, Schiowitz B, Rait G, Vickerstaff V, Sampson EL. Davies N, et al. Int Psychogeriatr. 2019 Oct;31(10):1403-1419. doi: 10.1017/S1041610219000826. Int Psychogeriatr. 2019. PMID: 31347482
- Resources to Support Decision-Making Regarding End-of-Life Nutrition Care in Long-Term Care: A Scoping Review. Alford H, Anvari N, Lengyel C, Wickson-Griffiths A, Hunter P, Yakiwchuk E, Cammer A. Alford H, et al. Nutrients. 2024 Apr 13;16(8):1163. doi: 10.3390/nu16081163. Nutrients. 2024. PMID: 38674853 Free PMC article. Review.
- The impact of the mySupport advance care planning intervention on family caregivers' perceptions of decision-making and care for nursing home residents with dementia: pretest-posttest study in six countries. Bavelaar L, Visser M, Walshe C, Preston N, Kaasalainen S, Sussman T, Cornally N, Hartigan I, Loucka M, di Giulio P, Brazil K, Achterberg WP, van der Steen JT. Bavelaar L, et al. Age Ageing. 2023 Mar 1;52(3):afad027. doi: 10.1093/ageing/afad027. Age Ageing. 2023. PMID: 36861181 Free PMC article.
- Support needs of carers making proxy healthcare decisions for people with dementia: a systematic review based on the Noblit and Hare meta-ethnographic synthesis of qualitative studies. Hodges V, Hynes C, Lassa S, Mitchell C. Hodges V, et al. BMJ Open. 2021 Dec 20;11(12):e052608. doi: 10.1136/bmjopen-2021-052608. BMJ Open. 2021. PMID: 34930737 Free PMC article.
- Caregiving for Patients With Frontotemporal Dementia in Latin America. Piña-Escudero SD, Aguirre GA, Javandel S, Longoria-Ibarrola EM. Piña-Escudero SD, et al. Front Neurol. 2021 Jul 7;12:665694. doi: 10.3389/fneur.2021.665694. eCollection 2021. Front Neurol. 2021. PMID: 34305781 Free PMC article.
- Alzheimer’s Association. 2016 Alzheimer’s disease facts and figures. Alzheimers Dement 2016; 12(4): 459–509. - PubMed
- Hanson LC, Carey TS, Caprio AJ, et al. Improving decision-making for feeding options in advanced dementia: a randomized, controlled trial. J Am Geriatr Soc 2011; 59(11): 2009–2016. - PMC - PubMed
- De la Cuesta-Benjumea C. The legitimacy of rest: conditions for the relief of burden in advanced dementia care-giving. J Adv Nurs 2010; 66(5): 988–998. - PubMed
- National Academies of Sciences, Engineering, and Medicine. Families caring for an aging America. Washington, DC: The National Academies Press, 2016. - PubMed
- Givens JL, Kiely DK, Carey K, et al. Healthcare proxies of nursing home residents with advanced dementia: decisions they confront and their satisfaction with decision-making. J Am Geriatr Soc 2009; 57(7): 1149–1155. - PMC - PubMed
Publication types
- Search in MeSH
Related information
Linkout - more resources, full text sources.
- Europe PubMed Central
- PubMed Central
Other Literature Sources
- scite Smart Citations
Miscellaneous
- NCI CPTAC Assay Portal

- Citation Manager
NCBI Literature Resources
MeSH PMC Bookshelf Disclaimer
The PubMed wordmark and PubMed logo are registered trademarks of the U.S. Department of Health and Human Services (HHS). Unauthorized use of these marks is strictly prohibited.
Information
- Author Services
Initiatives
You are accessing a machine-readable page. In order to be human-readable, please install an RSS reader.
All articles published by MDPI are made immediately available worldwide under an open access license. No special permission is required to reuse all or part of the article published by MDPI, including figures and tables. For articles published under an open access Creative Common CC BY license, any part of the article may be reused without permission provided that the original article is clearly cited. For more information, please refer to https://www.mdpi.com/openaccess .
Feature papers represent the most advanced research with significant potential for high impact in the field. A Feature Paper should be a substantial original Article that involves several techniques or approaches, provides an outlook for future research directions and describes possible research applications.
Feature papers are submitted upon individual invitation or recommendation by the scientific editors and must receive positive feedback from the reviewers.
Editor’s Choice articles are based on recommendations by the scientific editors of MDPI journals from around the world. Editors select a small number of articles recently published in the journal that they believe will be particularly interesting to readers, or important in the respective research area. The aim is to provide a snapshot of some of the most exciting work published in the various research areas of the journal.
Original Submission Date Received: .
- Active Journals
- Find a Journal
- Proceedings Series
- For Authors
- For Reviewers
- For Editors
- For Librarians
- For Publishers
- For Societies
- For Conference Organizers
- Open Access Policy
- Institutional Open Access Program
- Special Issues Guidelines
- Editorial Process
- Research and Publication Ethics
- Article Processing Charges
- Testimonials
- Preprints.org
- SciProfiles
- Encyclopedia

Article Menu

- Subscribe SciFeed
- Recommended Articles
- Google Scholar
- on Google Scholar
- Table of Contents
Find support for a specific problem in the support section of our website.
Please let us know what you think of our products and services.
Visit our dedicated information section to learn more about MDPI.
JSmol Viewer
Critical factors driving construction project performance in integrated 5d building information modeling.

1. Introduction
2. materials and methods, 2.1. procedure for the systematic literature review, 2.2. search strategy, 2.3. tools and software, 2.4. data resources, 2.5. prisma flow for systematic review, 2.6. network analysis, 3.1. development trends for 5d bim in the construction industry, 3.1.1. publications per year, 3.1.2. major countries or regions undertaking research, 3.1.3. key productive authors, 3.1.4. keyword co-occurrence and cluster identification, 3.2. factors significantly affecting the adoption of 5d bim, 3.3. key project performance factors affected by the implementation of 5d bim, 4. discussion, 4.1. evolution of 5d bim in the construction industry, 4.1.1. citation bursts and trend evaluation, 4.1.2. cluster analysis, 4.2. critical factors influencing the implementation of 5d bim, 4.2.1. technology factors, 4.2.2. organizational factors, 4.2.3. environmental factors, 4.2.4. operator factors, 4.2.5. project factors, 4.2.6. government policy, 4.3. key performance indicators affected by the implementation of 5d bim, 4.3.1. project cost performance, 4.3.2. project time performance, 4.3.3. project quality performance, 5. limitations and future research directions, 6. conclusions, author contributions, data availability statement, conflicts of interest.
- Abubakar, A.S.; Haron, N.A.; Alias, A.H.; Teik Hua, L. Exploring quality dimensions from a construction perspective: A literature review. J. Teknol. 2023 , 85 , 133–141. [ Google Scholar ] [ CrossRef ]
- Hofstadler, C.; Kummer, M. Influence of project preparation quality on the project outcome: A qualitative approach. In Proceedings of the 10th International Structural Engineering and Construction Conference, Chicago, IL, USA, 20–25 May 2019; Volume 6. [ Google Scholar ] [ CrossRef ]
- Anoop, T.; Asadi, S.; Prasad, A.V.S. The critical success factors affecting the performance of construction industry. Int. J. Civ. Eng. Technol. 2016 , 7 , 669–675. [ Google Scholar ]
- Wong, P.F.; Salleh, H.; Rahim, F.A.M. A Relationship Framework for Building Information Modeling (BIM) Capability in Quantity Surveying Practice and Project Performance. Inf. Constr. 2015 , 67 , e119. [ Google Scholar ] [ CrossRef ]
- Emadi, S.; Emadi, S. Analyzing Cost and Time Objectives in the Construction Projects Using Artificial Neural Network. IRECE 2022 , 13 , 91. [ Google Scholar ] [ CrossRef ]
- Cavalline, T.L.; Morian, D.A.; Schexnayder, C.J. Construction Quality in the Alternate Project Delivery Environment ; American Society of Civil Engineers (ASCE): Reston, VA, USA, 2021; p. 446. ISBN 978-078448363-3. [ Google Scholar ]
- Jaafar, K.; Watfa, M. A Multi-Objective Optimization Approach for the Cost-Time-Quality Trade-Off in Construction Projects. J. Mod. Proj. Manag. 2021 , 10 , 187–197. [ Google Scholar ]
- Galjanić, K.; Marović, I.; Jajac, N. Decision Support Systems for Managing Construction Projects: A Scientific Evolution Analysis. Sustainability 2022 , 14 , 4977. [ Google Scholar ] [ CrossRef ]
- Luo, L.; He, Q.; Jaselskis, E.J.; Xie, J. Construction Project Complexity: Research Trends and Implications. J. Constr. Eng. Manag. 2017 , 143 , 04017019. [ Google Scholar ] [ CrossRef ]
- Zhu, F.; Wang, X.; Wang, L.; Yu, M. Project Manager’s Emotional Intelligence and Project Performance: The Mediating Role of Project Commitment. Int. J. Proj. Manag. 2021 , 39 , 788–798. [ Google Scholar ] [ CrossRef ]
- Vasiliev, A.; Bruskin, S. Key Indicators, Innovations, Trends, and Main Factors Influencing Transformation of the Building Construction Industry in Russia. In Digitalization of Society, Economics and Management ; Springer International Publishing: Berlin/Heidelberg, Germany, 2022; Volume 53, pp. 15–28. [ Google Scholar ]
- Ershadi, M.; Lijauco, F. A Framework for Digitalizing Construction Businesses: Drivers, Barriers and Outcomes. Smart Sustain. Built Environ. 2024 , 13 . [ Google Scholar ] [ CrossRef ]
- Sezer, A.A.; Thunberg, M.; Wernicke, B. Digitalization Index: Developing a Model for Assessing the Degree of Digitalization of Construction Projects. J. Constr. Eng. Manag. 2021 , 147 , 04021119. [ Google Scholar ] [ CrossRef ]
- Stoyanova, M. Good Practices and Recommendations for Success in Construction Digitalization. TEM J. 2020 , 9 , 42–47. [ Google Scholar ] [ CrossRef ]
- Marhraoui, M.A. Digital Skills for Project Managers: A Systematic Literature Review. Procedia Comput. Sci. 2023 , 219 , 1591–1598. [ Google Scholar ] [ CrossRef ]
- Bosch-Sijtsema, P.; Claeson-Jonsson, C.; Johansson, M.; Roupe, M. The Hype Factor of Digital Technologies in AEC. CI 2021 , 21 , 899–916. [ Google Scholar ] [ CrossRef ]
- Tatjana, B. The Application of BIM Technology and Its Reliability in the Static Load Analysis. Teh. Vjesn. 2016 , 23 , 1221–1226. [ Google Scholar ] [ CrossRef ]
- Ahmed, Y.A.; Shehzad, H.M.F.; Khurshid, M.M.; Abbas Hassan, O.H.; Abdalla, S.A.; Alrefai, N. Examining the Effect of Interoperability Factors on Building Information Modelling (BIM) Adoption in Malaysia. Constr. Innov. 2024 , 24 , 606–642. [ Google Scholar ] [ CrossRef ]
- Platonov, A.; Larionova, V.; Davy, Y. Development of Approaches and Organizational Models for the Mass Implementation of Information Modeling Technologies in the Investment and Construction Sphere. J. Risk. Financ. Manag. 2023 , 16 , 118. [ Google Scholar ] [ CrossRef ]
- Pakhale, P.D.; Pal, A. Digital Project Management in Infrastructure Project: A Case Study of Nagpur Metro Rail Project. Asian J. Civ. Eng. 2020 , 21 , 639–647. [ Google Scholar ] [ CrossRef ]
- Hosamo, H.H.; Rolfsen, C.N.; Zeka, F.; Sandbeck, S.; Said, S.; Sætre, M.A. Navigating the Adoption of 5D Building Information Modeling: Insights from Norway. Infrastructures 2024 , 9 , 75. [ Google Scholar ] [ CrossRef ]
- Pan, H.; Pan, Y.; Wu, C. Research on Integrated Management of L Hotel Construction Project Based on BIM5D. In Proceedings of the ICCREM 2022, American Society of Civil Engineers, Virtual, 15 December 2022; pp. 461–469. [ Google Scholar ]
- Xu, J. Research on Application of BIM 5D Technology in Central Grand Project. Procedia Eng. 2017 , 174 , 600–610. [ Google Scholar ] [ CrossRef ]
- MacFeely, S. Measuring the Sustainable Development Goal Indicators: An Unprecedented Statistical Challenge. J. Off. Stat. 2020 , 36 , 361–378. [ Google Scholar ] [ CrossRef ]
- Abdelwahab, M.A.; Nassar, A.H. Legal Barriers and Associated Solutions of BIM Implementation in the Egyptian AEC Industry. Int. J. Constr. Manag. 2023 , 24 , 1431–1443. [ Google Scholar ] [ CrossRef ]
- Belay, S.; Goedert, J.; Woldesenbet, A.; Rokooei, S. Enhancing BIM Implementation in the Ethiopian Public Construction Sector: An Empirical Study. Cogent Eng. 2021 , 8 , 1886476. [ Google Scholar ] [ CrossRef ]
- Chan, D.W.M.; Olawumi, T.O.; Ho, A.M.L. Perceived Benefits of and Barriers to Building Information Modelling (BIM) Implementation in Construction: The Case of Hong Kong. J. Build. Eng. 2019 , 25 , 100764. [ Google Scholar ] [ CrossRef ]
- Tai, S.; Zhang, Y.; Li, T. Factors Affecting BIM Application in China: A Social Network Model. JEDT 2021 , 19 , 373–384. [ Google Scholar ] [ CrossRef ]
- Olsen, D.; Taylor, J.M. Quantity Take-Off Using Building Information Modeling (BIM), and Its Limiting Factors. Procedia Eng. 2017 , 196 , 1098–1105. [ Google Scholar ] [ CrossRef ]
- Mesároš, P.; Smetanková, J.; Mandičák, T. The Fifth Dimension of BIM—Implementation Survey. In IOP Conference Series: Earth and Environmental Science ; IOP Publishing: Bristol, UK, 2019; Volume 222. [ Google Scholar ]
- Hashim, M.Z.; Othman, I.; Khalid, N.S.M.; Hassan, S.H.; Musa, M.K. Construction Players’ Awareness on the Use of Building Information Modelling (BIM) and Industrialized Building System (IBS) in Malaysian Construction Industry. In Proceedings of the International Conference on Emerging Smart Cities (ICESC2022), Kuching, Malaysia, 1–2 December 2022; Springer Nature: Singapore, 2024; Volume 324, pp. 491–504. [ Google Scholar ]
- Arellano, K.; Castillo, T. Assessment of BIM Use in the Early Stages of Implementation Evaluación Del Uso de BIM En Las Primeras Fases de Aplicación. Rev. Ing. Constr. 2021 , 36 , 311–321. [ Google Scholar ] [ CrossRef ]
- Sattineni, A.; Macdonald, J.A. 5D-BIM: A case study of an implementation strategy in the construction industry. In ISARC. Proceedings of the International Symposium on Automation and Robotics in Construction ; IAARC Publications: Sydney, Australia, 2014. [ Google Scholar ]
- Stanley, R.; Thurnell, D. The Benefits of, and Barriers to, Implementation of 5D BIM for Quantity Surveying in New Zealand. Australas. J. Constr. Econ. Build. 2014 , 14 , 105–117. [ Google Scholar ] [ CrossRef ]
- Thurairajah, N.; Goucher, D. Advantages and Challenges of Using BIM: A Cost Consultant’s Perspective. In Proceedings of the 49th ASC Annual International Conference, San Luis Obispo, CA, USA, 10–13 April 2013. [ Google Scholar ]
- Hasan, A.N.; Rasheed, S.M. The Benefits of and Challenges to Implement 5D BIM in Construction Industry. Civ. Eng. J. 2019 , 5 , 412–421. [ Google Scholar ] [ CrossRef ]
- Harrison, C.; Thurnell, D. BIM Implementation in a New Zealand Consulting Quantity Surveying Practice. IJCSCM 2015 , 5 , 1–15. [ Google Scholar ] [ CrossRef ]
- Girginkaya Akdag, S.; Maqsood, U. A Roadmap for BIM Adoption and Implementation in Developing Countries: The Pakistan Case. Archnet-IJAR 2020 , 14 , 112–132. [ Google Scholar ] [ CrossRef ]
- Alhasan, S.; Kumar, B.; Thanikal, J.V. Effectiveness of Implementing 5D Functions of Building Information Modeling on Professions of Quantity Surveying—A Review. Int.J. Civ. Eng. Technol. 2017 , 8 , 783–800. [ Google Scholar ]
- Ahmad, A.; Sarip, S.; Fathi, M.S.; Abd Fatah, A.Y. Application of 5D BIM to Improve the Oil and Gas Project Cost Performance. In AIP Conference Proceedings ; American Institute of Physics Inc.: College Park, MD, USA, 2023; Volume 2827. [ Google Scholar ]
- Moses, T.; Heesom, D.; Oloke, D. Implementing 5D BIM on Construction Projects: Contractor Perspectives from the UK Construction Sector. J. Eng. Des. Technol. 2020 , 18 , 1867–1888. [ Google Scholar ] [ CrossRef ]
- Aladağ, H.; Demirdöğen, G.; Demirbağ, A.T.; Işık, Z. Understanding the Perception Differences on BIM Adoption Factors across the Professions of AEC Industry. Ain Shams Eng. J. 2023 , 14 , 102545. [ Google Scholar ] [ CrossRef ]
- Van Tam, N.; Diep, T.N.; Quoc Toan, N.; Le Dinh Quy, N. Factors Affecting Adoption of Building Information Modeling in Construction Projects: A Case of Vietnam. Cogent Bus. Manag. 2021 , 8 , 1918848. [ Google Scholar ] [ CrossRef ]
- Abbas, S.K.; Ansari, U.A.; Ahmed, B.; Asif, J. Identify the Effect of Awareness Upon Mutual Funds Investor Base in Pakistan Using Moderation. Eurasian J. Anal. Chem. 2018 , 13 , 224–233. [ Google Scholar ]
- Moradi, S.; Ansari, R.; Taherkhani, R. A Systematic Analysis of Construction Performance Management: Key Performance Indicators from 2000 to 2020. Iran. J. Sci. Technol. Trans. Civ. Eng. 2022 , 46 , 15–31. [ Google Scholar ] [ CrossRef ]
- Mutia Hanuun Ufaira, A.; Yusuf, Y. Risks in the Use of BIM 5D in the Estimated Cost of the Project Tender Phase. In AIP Conference Proceedings ; AIP Publishing: College Park, MD, USA, 2024; pp. 020118-1–020118-6. [ Google Scholar ]
- Herdyana, H.; Suroso, A. The Impacts of 5D-Building Information Modeling on Construction’s Time and Cost Performance. Sinergi 2023 , 27 , 393–404. [ Google Scholar ] [ CrossRef ]
- Kunkcu, H.; Koc, K.; Dagou, H.H.; Gurgun, A.P. Using key performance indicators in construction project literature. Proc. Int. Struct. Eng. Constr. 2022 , 9 , 1–6. [ Google Scholar ] [ CrossRef ]
- Ngacho, C.; Das, D. A Performance Evaluation Framework of Construction Projects: Insights from Literature. IJPOM 2015 , 7 , 151. [ Google Scholar ] [ CrossRef ]
- Chan, A.P.C.; Chan, A.P.L. Key Performance Indicators for Measuring Construction Success. Benchmarking Int. J. 2004 , 11 , 203–221. [ Google Scholar ] [ CrossRef ]
- Wang, T.; Abdallah, M.; Clevenger, C.; Monghasemi, S. Time–Cost–Quality Trade-off Analysis for Planning Construction Projects. ECAM 2021 , 28 , 82–100. [ Google Scholar ] [ CrossRef ]
- Hashim Yas, A.; Neamah Ahmed, M.; Abdulhadi, A.M. Decision Support System Based on BWM for Analyzing Success Factors Affecting the Quality in the Iraqi Construction Projects. PEN 2023 , 11 , 131. [ Google Scholar ] [ CrossRef ]
- Albtoush, A.M.F.; Doh, S.I.; Rahman, R.A.; Al-Momani, A.H. Critical Success Factors of Construction Projects in Jordan: An Empirical Investigation. Asian J. Civ. Eng. 2022 , 23 , 1087–1099. [ Google Scholar ] [ CrossRef ]
- Hu, W.; He, X. An Innovative Time-Cost-Quality Tradeoff Modeling of Building Construction Project Based on Resource Allocation. Sci. World J. 2014 , 2014 , 673248. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Rajabi, M.S.; Radzi, A.R.; Rezaeiashtiani, M.; Famili, A.; Rashidi, M.E.; Rahman, R.A. Key Assessment Criteria for Organizational BIM Capabilities: A Cross-Regional Study. Buildings 2022 , 12 , 1013. [ Google Scholar ] [ CrossRef ]
- Adriaanse, A.; Voordijk, H.; Dewulf, G. The Use of Interorganisational ICT in United States Construction Projects. Autom. Constr. 2010 , 19 , 73–83. [ Google Scholar ] [ CrossRef ]
- Park, H.; Choi, S.O. Digital Innovation Adoption and Its Economic Impact Focused on Path Analysis at National Level. J. Open Innov. Technol. Mark. Complex. 2019 , 5 , 56. [ Google Scholar ] [ CrossRef ]
- Krüger, J.; Lausberger, C.; Von Nostitz-Wallwitz, I.; Saake, G.; Leich, T. Search. Review. Repeat? An empirical study of threats to replicating SLR searches. Empir. Softw. Eng. 2020 , 25 , 627–677. [ Google Scholar ] [ CrossRef ]
- Apóstolo, J.L.A.; Jla, A. Decoding the Synthesis of Evidence: Concepts. Online Braz. J. Nurs. 2016 , 15 , 336–340. [ Google Scholar ] [ CrossRef ]
- Wang, G.; Liu, Z.; Wang, H. Key Factors Affecting BIM Adoption Is China Based on TOEaRC. In Proceedings of the 2016 International Conference on Mechanics, Materials and Structural Engineering, Jeju Island, Republic of Korea, 18–20 March 2016; Atlantis Press: Amsterdam, The Netherlands, 2016. [ Google Scholar ]
- Tornatzky, L.G.; Fleischer, M.; Chakrabarti, A.K. The Processes of Technological Innovation ; Lexington Books: Lanham, MD, USA, 1990; ISBN 0-669-20348-3. [ Google Scholar ]
- Sun, Y.; Tan, C.; Lim, K.H.; Liang, T.; Yeh, Y. Strategic Contexts, Strategic Orientations and Organisational Technology Adoption: A Configurational Approach. Inf. Syst. J. 2024 , 34 , 1355–1401. [ Google Scholar ] [ CrossRef ]
- Creswell, J.W. Research Design: Qualitative, Quantitative, and Mixed Methods Approaches , 3rd ed.; Sage Publications: Thousand Oaks, CA, USA, 2009; ISBN 978-1-4129-6556-9. [ Google Scholar ]
- Purushothaman, M.B.; Seadon, J. System-Wide Construction Waste and Their Connectivity to Construction Phases, Impacting 5M Factors and Effects: A Systematic Review. Smart Sustain. Built Environ. 2024 , 13 , 354–369. [ Google Scholar ] [ CrossRef ]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A. Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols (PRISMA-P) 2015 Statement. Syst. Rev. 2015 , 4 , 1. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Pawson, R.; Greenhalgh, T.; Harvey, G.; Walshe, K. Realist Review—A New Method of Systematic Review Designed for Complex Policy Interventions. J. Health Serv. Res. Policy 2005 , 10 , 21–34. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- PRISMA 2020. Available online: https://www.prisma-statement.org/ (accessed on 1 March 2024).
- Van Eck, N.J.; Waltman, L. Software Survey: VOSviewer, a Computer Program for Bibliometric Mapping. Scientometrics 2010 , 84 , 523–538. [ Google Scholar ] [ CrossRef ]
- Yang, M.; Zhao, S.; Zeng, W. The Analysis of Knowledge Base, Theme Evolution and Research Hotspot of Ideological and Political Theory Course in Colleges and Universities Based on CiteSpace. In Proceedings of the 2021 2nd International Conference on Artificial Intelligence and Education (ICAIE), Dali, China, 18–20 June 2021; IEEE: Piscataway, NJ, USA, 2021; pp. 460–467. [ Google Scholar ]
- Zhao, Y.; Taib, N. Cloud-Based Building Information Modelling (Cloud-BIM): Systematic Literature Review and Bibliometric-Qualitative Analysis. Autom. Constr. 2022 , 142 , 104468. [ Google Scholar ] [ CrossRef ]
- Singh, V.K.; Singh, P.; Karmakar, M.; Leta, J.; Mayr, P. The Journal Coverage of Web of Science, Scopus and Dimensions: A Comparative Analysis. Scientometrics 2021 , 126 , 5113–5142. [ Google Scholar ] [ CrossRef ]
- Birkle, C.; Pendlebury, D.A.; Schnell, J.; Adams, J. Web of Science as a Data Source for Research on Scientific and Scholarly Activity. Quant. Sci. Stud. 2020 , 1 , 363–376. [ Google Scholar ] [ CrossRef ]
- Codina, L.; Morales-Vargas, A.; Rodríguez-Martínez, R.; Pérez-Montoro, M. Uso de Scopus y Web of Science para investigar y evaluar en comunicación social: Análisis comparativo y caracterización. Index Comun. 2020 , 10 , 235–261. [ Google Scholar ] [ CrossRef ]
- Osei-Kyei, R.; Narbaev, T.; Ampratwum, G. A Scientometric Analysis of Studies on Risk Management in Construction Projects. Buildings 2022 , 12 , 1342. [ Google Scholar ] [ CrossRef ]
- Padwiansyah, M.R.; Wahyuningsih, R.S.H.; Handayani, S.D. Analyzing Sustainable Organizational Development with VOSviewer: A Bibliometric Analysis. Multidiscip. Rev. 2023 , 6 , 2023023. [ Google Scholar ] [ CrossRef ]
- Alnajem, M.; Mostafa, M.M.; ElMelegy, A.R. Mapping the First Decade of Circular Economy Research: A Bibliometric Network Analysis. J. Ind. Prod. Eng. 2021 , 38 , 29–50. [ Google Scholar ] [ CrossRef ]
- Samad, S.A.; Harun, A.N.; Nawi, M.N.M.; Haron, N.A. The potential use of BIM through an electronic submission: A preliminary study. Malays. Constr. Res. J. 2018 , 3 , 82–96. [ Google Scholar ]
- Abdel-Hamid, M.; Abdelhaleem, H.M. Project Cost Control Using Five Dimensions Building Information Modelling. Int. J. Constr. Manag. 2023 , 23 , 405–409. [ Google Scholar ] [ CrossRef ]
- Smith, P. Project Cost Management with 5D BIM. Procedia Soc. Behav. Sci. 2016 , 226 , 193–200. [ Google Scholar ] [ CrossRef ]
- Mayouf, M.; Gerges, M.; Cox, S. 5D BIM: An Investigation into the Integration of Quantity Surveyors within the BIM Process. J. Eng. Des. Technol. 2019 , 17 , 537–553. [ Google Scholar ] [ CrossRef ]
- Aibinu, A.; Venkatesh, S. Status of BIM Adoption and the BIM Experience of Cost Consultants in Australia. J. Prof. Issues Eng. Educ. Pract. 2014 , 140 , 04013021. [ Google Scholar ] [ CrossRef ]
- Abdullahi, A.M.; Mohamed, S.F.; Misnan, M.S.; Abanda, F.H.; Abdulrahman, F.H. Recent Advances in 5D BIM Cost Control: A Novel Ontological Approach. In Proceedings of the Thirteenth International Conference on Construction in the 21st Century (CITC-13), Arnhem, The Netherlands, 8–11 May 2023. [ Google Scholar ]
- Smith, P. BIM & the 5D Project Cost Manager. Procedia—Soc. Behav. Sci. 2014 , 119 , 475–484. [ Google Scholar ]
- Kapogiannis, G.; Gaterell, M.; Oulasoglou, E. Identifying Uncertainties Toward Sustainable Projects. Procedia Eng. 2015 , 118 , 1077–1085. [ Google Scholar ] [ CrossRef ]
- Sun, H.; Khoo, T.J.; Ha, C.Y.; Chen, S.; Deng, Z.; Shi, Y. The Impacts of 5D Building Information Modelling towards Cost Management in the Construction Industry. J. Adv. Res. Appl. Sci. Eng. Technol. 2023 , 32 , 471–487. [ Google Scholar ] [ CrossRef ]
- Chen, C.; Tang, L.C.M.; Jin, Y. Development of 5D Bim-Based Management System for Pre-Fabricated Construction in China. In Proceedings of the International Conference on Smart Infrastructure and Construction 2019 (ICSIC) Driving Data-Informed Decision-Making, Cambridge, UK, 8–10 July 2019; ICE Publishing: Lancashire, UK, 2019; pp. 215–224. [ Google Scholar ]
- Ingle, P.V.; Mahesh, G. Construction Project Performance Areas for Indian Construction Projects. Int. J. Constr. Manag. 2022 , 22 , 1443–1454. [ Google Scholar ] [ CrossRef ]
- Ofori, G. Get Construction Project Performance Parameters Right to Attain Sustainable Development Goals. Sustainability 2023 , 15 , 13360. [ Google Scholar ] [ CrossRef ]
- Famiyeh, S.; Amoatey, C.T.; Adaku, E.; Agbenohevi, C.S. Major Causes of Construction Time and Cost Overruns: A Case of Selected Educational Sector Projects in Ghana. JEDT 2017 , 15 , 181–198. [ Google Scholar ] [ CrossRef ]
- Mering, M.M.; Aminudin, E.; Chai, C.S.; Zakaria, R.; Tan, C.S.; Lee, Y.Y.; Redzuan, A.A. Adoption of Building Information Modelling in Project Planning Risk Management. In IOP Conference Series: Materials Science and Engineering ; Institute of Physics Publishing: Bristol, UK, 2017; Volume 271. [ Google Scholar ]
- Vigneault, M.-A.; Boton, C.; Chong, H.-Y.; Cooper-Cooke, B. An Innovative Framework of 5D BIM Solutions for Construction Cost Management: A Systematic Review. Arch. Comput. Methods Eng. 2020 , 27 , 1013–1030. [ Google Scholar ] [ CrossRef ]
- Wang, X.; Yung, P.; Luo, H.; Truijens, M. An Innovative Method for Project Control in LNG Project through 5D CAD: A Case Study. Autom. Constr. 2014 , 45 , 126–135. [ Google Scholar ] [ CrossRef ]
- Gohil, P.P.; Malek, M.; Bachwani, D.; Patel, D.; Upadhyaya, D.; Hathiwala, A. Application of 5D Building Information Modeling for Construction Management. ECS Trans. 2022 , 107 , 2637–2649. [ Google Scholar ] [ CrossRef ]
- Hussain, O.A.I.; Moehler, R.C.; Walsh, S.D.C.; Ahiaga-Dagbui, D.D. Minimizing Cost Overrun in Rail Projects through 5D-BIM: A Systematic Literature Review. Infrastructures 2023 , 8 , 93. [ Google Scholar ] [ CrossRef ]
- Leicht, D.; Castro-Fresno, D.; Diaz, J.; Baier, C. Multidimensional Construction Planning and Agile Organized Project Execution-The 5D-PROMPT Method. Sustainability 2020 , 12 , 6340. [ Google Scholar ] [ CrossRef ]
- Nast, A.; Koch, C. Practical Experiences with 5D Building Information Modeling—A Systematic Literature Review. In Proceedings of the ECPPM 2022-eWork and eBusiness in Architecture, Engineering and Construction 2022, Trondheim, Norway, 14–16 September 2022; Hjelseth, E., Sujan, S.F., Scherer, R.J., Eds.; CRC Press: Boca Raton, FL, USA, 2023; pp. 508–515. [ Google Scholar ]
- Azhar, S. Building Information Modeling (BIM): Trends, Benefits, Risks, and Challenges for the AEC Industry. Leadersh. Manag. Eng. 2011 , 11 , 241–252. [ Google Scholar ] [ CrossRef ]
- Choi, H.; Zo, H. Assessing the Efficiency of National Innovation Systems in Developing Countries. Sci. Public Policy 2019 , 46 , 530–540. [ Google Scholar ] [ CrossRef ]
- Hu, X.; Chen, Y. Impact of BIM on Project Performance. In Proceedings of the EC3 Conference 2023 European Council on Computing in Construction, Crete, Greece, 10 July 2023. [ Google Scholar ]
- Rohani, M.; Shafabakhsh, G.; Haddad, A.; Asnaashari, E. Strategy Management of Construction Workspaces by Conflict Resolution Algorithm and Visualization Model. Eng. Constr. Archit. Manag. 2018 , 25 , 1053–1074. [ Google Scholar ] [ CrossRef ]
- Lu, Q.; Won, J.; Cheng, J.C.P. A Financial Decision Making Framework for Construction Projects Based on 5D Building Information Modeling (BIM). Int. J. Proj. Manag. 2016 , 34 , 3–21. [ Google Scholar ] [ CrossRef ]
- Scheer, S.; Mendes, R.; Campestrini, T.F.; Garrido, M.C. On-Site BIM Model Use to Integrate 4D/5D Activities and Construction Works: A Case Study on a Brazilian Low Income Housing Enterprise. Comput. Civ. Build. Eng. 2014 , 2014 , 455–462. [ Google Scholar ]
- ElQasaby, A.R.; Alqahtani, F.K.; Alheyf, M. Automated Schedule and Cost Control Using 3D Sensing Technologies. Appl. Sci. 2023 , 13 , 783. [ Google Scholar ] [ CrossRef ]
- Agostinelli, S.; Cinquepalmi, F.; Ruperto, F. 5d Bim: Tools and Methods for Digital Project Construction Management. WIT Trans. Built Environ. 2019 , 192 , 205–215. [ Google Scholar ]
- Nicolas, S.; Daniel, F.; Conrad, B. An Interactive Decision-Support Tool to Improve Construction Cost Management with Building Information Modeling. In Proceedings of the Canadian Society of Civil Engineering Annual Conference; Springer: Berlin/Heidelberg, Germany, 2023; Volume 363, pp. 687–698. [ Google Scholar ]
- Kang, L.S.; Kim, H.S.; Moon, H.S.; Kim, S.-K. Managing Construction Schedule by Telepresence: Integration of Site Video Feed with an Active nD CAD Simulation. Autom. Constr. 2016 , 68 , 32–43. [ Google Scholar ] [ CrossRef ]
- Ybañez, R.S.; De La Cruz, A.R. Related Literature Review 5D Model for Project and Operation/Maintenance Remote Monitoring of Equipment and Piping System. JESA 2023 , 56 , 355–364. [ Google Scholar ] [ CrossRef ]
- He, C.; Hou, Y.; Ding, L.; Li, P. Visualized Literature Review on Sustainable Building Renovation. J. Build. Eng. 2021 , 44 , 102622. [ Google Scholar ] [ CrossRef ]
- Chen, Z.-S.; Yang, Y.; Wang, X.-J.; Chin, K.-S.; Tsui, K.-L. Fostering Linguistic Decision-Making under Uncertainty: A Proportional Interval Type-2 Hesitant Fuzzy TOPSIS Approach Based on Hamacher Aggregation Operators and Andness Optimization Models. Inf. Sci. 2019 , 500 , 229–258. [ Google Scholar ] [ CrossRef ]
- Chen, C.; Ibekwe-SanJuan, F.; Hou, J. The Structure and Dynamics of Cocitation Clusters: A Multiple-perspective Cocitation Analysis. J. Am. Soc. Inf. Sci. 2010 , 61 , 1386–1409. [ Google Scholar ] [ CrossRef ]
- Chai, C.; Mustafa, K.; Kuppusamy, S.; Yusof, A.; Lim, C.S.; Wai, S.H. BIM Integration in Augmented Reality Model. IJTech 2019 , 10 , 1266. [ Google Scholar ] [ CrossRef ]
- Wu, W.; Issa, R.R.A. Key Issues in Workforce Planning and Adaptation Strategies for BIM Implementation in Construction Industry. In Proceedings of the Construction Research Congress 2014, American Society of Civil Engineers, Atlanta, GA, USA, 13 May 2014; pp. 847–856. [ Google Scholar ]
- Wu, Q.; Chen, L.; Shi, P.; Wang, W.; Xu, S. Identifying Impact Factors of MEP Installation Productivity: An Empirical Study. Buildings 2022 , 12 , 565. [ Google Scholar ] [ CrossRef ]
- Awwad, K.A.; Shibani, A.; Ghostin, M. Exploring the Critical Success Factors Influencing BIM Level 2 Implementation in the UK Construction Industry: The Case of SMEs. Int. J. Constr. Manag. 2022 , 22 , 1894–1901. [ Google Scholar ] [ CrossRef ]
- Belay, S.; Goedert, J.; Woldesenbet, A.; Rokooei, S.; Matos, J. Building Information Modeling Implementation Strategies for Public Infrastructure Projects in Emerging Markets: The Case of Ethiopia. Cogent Eng. 2023 , 10 , 2220481. [ Google Scholar ] [ CrossRef ]
- Muthusamy, K.; Chew, L. Critical Success Factors for the Implementation of Building Information Modeling (BIM) among Construction Industry Development Board (CIDB) G7 Contractors in the Klang Valley, Malaysia. In Proceedings of the 2020 IEEE European Technology and Engineering Management Summit (E-TEMS), Dortmund, Germany, 5–7 March 2020; IEEE: Piscataway, NJ, USA, 2020; pp. 1–6. [ Google Scholar ]
- Chulok, A.; Kotsemir, M.; Radomirova, Y.; Shashnov, S. Setting S&T Perspective Priorities in an Era of Complexity and Uncertainty: A Case for Megacities. Foresight 2024 , 26 , 365–391. [ Google Scholar ] [ CrossRef ]
- Rusnák, J.; Ďurček, P.; Korec, P.; Plešivčák, M. Questing for Contexts in the Research of Regional Inequalities in Slovakia: Application of the Concept of Critical Realism. Acta Geogr. Univ. Comen. 2023 , 67 , 187–215. [ Google Scholar ]
- Zhao, S.L.; Zhu, D.Y.; Peng, X.B.; Song, W. An Empirical Analysis of the Regional Competitiveness Based on S&T Talents Flow. Hum. Syst. Manag. 2016 , 35 , 1–10. [ Google Scholar ] [ CrossRef ]
- Yao, Y.; Qin, H.; Wang, J. Barriers of 5D BIM Implementation in Prefabrication Construction of Buildings. In IOP Conference Series: Earth and Environmental Science ; IOP Publishing Ltd.: Bristol, UK, 2020; Volume 580. [ Google Scholar ]
- Calado, M.P.; Ramos, A.; Fabiano, D. eHealth in Hospital Information Management. In Proceedings of the 2019 IST-Africa Week Conference (IST-Africa), Nairobi, Kenya, 8–10 May 2019; IEEE: Piscataway, NJ, USA, 2019; pp. 1–6. [ Google Scholar ]
- Kovacic, I.; Oberwinter, L.; Müller, C.; Achammer, C. The “BIM-Sustain” Experiment—Simulation of BIM-Supported Multi-Disciplinary Design. Vis. Eng. 2013 , 1 , 13. [ Google Scholar ] [ CrossRef ]
- Chowdhury, M.; Hosseini, M.R.; Edwards, D.J.; Martek, I.; Shuchi, S. Comprehensive Analysis of BIM Adoption: From Narrow Focus to Holistic Understanding. Autom. Constr. 2024 , 160 , 105301. [ Google Scholar ] [ CrossRef ]
- Hsiao, H.-C.; Chang, J.-C.; Chen, S.-C. The Influence of Support for Innovation on Organizational Innovation: Taking Organizational Learning as a Mediator. Asia-Pac. Edu Res. 2014 , 23 , 463–472. [ Google Scholar ] [ CrossRef ]
- Wangchuk, J.; Banihashemi, S.; Abbasianjahromi, H.; Antwi-Afari, M.F. Building Information Modelling in Hydropower Infrastructures: Design, Engineering and Management Perspectives. Infrastructures 2024 , 9 , 98. [ Google Scholar ] [ CrossRef ]
- Alnaser, A.A.; Hassan Ali, A.; Elmousalami, H.H.; Elyamany, A.; Gouda Mohamed, A. Assessment Framework for BIM-Digital Twin Readiness in the Construction Industry. Buildings 2024 , 14 , 268. [ Google Scholar ] [ CrossRef ]
- Olatunji, O.A.; Sher, W.; Ogunsemi, D.R. The Impact of Building Information Modelling on Construction Cost Estimation. In Proceedings of the W055-Special Track 18th CIB World Building Congress, Salford, UK, 10–13 May 2010; CIB Publication: Kanata, ON, Canada, 2010; pp. 193–201. [ Google Scholar ]
- Abualdenien, J.; Borrmann, A. Levels of Detail, Development, Definition, and Information Need: A Critical Literature Review. ITcon 2022 , 27 , 363–392. [ Google Scholar ] [ CrossRef ]
- Tan, S.; Gumusburun Ayalp, G. Root Factors Limiting BIM Implementation in Developing Countries: Sampling the Turkish AEC Industry. Open House INT 2022 , 47 , 732–762. [ Google Scholar ] [ CrossRef ]
- Rajabi, M.S.; Rezaeiashtiani, M.; Radzi, A.R.; Famili, A.; Rezaeiashtiani, A.; Rahman, R.A. Underlying Factors and Strategies for Organizational BIM Capabilities: The Case of Iran. ASI 2022 , 5 , 109. [ Google Scholar ] [ CrossRef ]
- Fan, S.-L. Intellectual Property Rights in Building Information Modeling Application in Taiwan. J. Constr. Eng. Manag. 2014 , 140 , 04013058. [ Google Scholar ] [ CrossRef ]
- Adibfar, A.; Costin, A.; Issa, R.R. Review of Copyright Challenges in AECO Industry and Prospective Developments for Building Information Modeling (BIM). In Proceedings of the Construction Research Congress 2020, American Society of Civil Engineers, Tempe, AZ, USA, 9 November 2020; pp. 1168–1176. [ Google Scholar ]
- Cheng, T.F.; Soon, L.K. Industry-Wide Adoption through a Comprehensive Building Information Modelling (BIM) Roadmap and BIM Electronic Regulatory Submission. In Proceedings of the Open Systems: 18th International Conference on Computer-Aided Architectural Design Research in Asia (CAADRIA 2013), Singapore, 15–18 May 2013; pp. 743–745. [ Google Scholar ]
- Masum, K.; Marma, B.; Islam, M.S.; Gazder, U. Fuzzy-Based Performance Measurement of Building Construction Projects: A Case Study. In Proceedings of the 7th IET Smart Cities Symposium (SCS 2023), Institution of Engineering and Technology, Hybrid Conference, Manama, Bahrain, 3–5 December 2023; pp. 23–26. [ Google Scholar ]
- Siddika, A.; Masfiqul Alam Bhuiyan, M. Methodological Analysis of KPIs to Evaluate Contractor Performance of the Construction Project. In Proceedings of the Canadian Society of Civil Engineering Annual Conference 2022; Springer Nature: Cham, Switzerland, 2024; Volume 367, pp. 639–652. [ Google Scholar ]
- Choi, K.; Yin, Y.; Goehl, D.; Jeong, H.D. Alternative Contracting Methods: Modeling and Assessing the Effects of Contract Type on Time-Cost-Change Performance. J. Manage. Eng. 2021 , 37 , 04020096. [ Google Scholar ] [ CrossRef ]
- Chicoca, D.; Utomo, C. Developers’ Perception of Cost Drivers Influencing Project Cost Performance on Surabaya High-Rise Residential, Indonesia. IOP Conf. Ser. Mater. Sci. Eng. 2019 , 650 , 012009. [ Google Scholar ] [ CrossRef ]
- Kim, B.-C. Probabilistic Evaluation of Cost Performance Stability in Earned Value Management. J. Manag. Eng. 2016 , 32 , 04015025. [ Google Scholar ] [ CrossRef ]
- Grau, D.; Back, W.E.; Abbaszadegan, A.; Sirven, R. The Predictability Index-a Novel Project Performance Metric to Assess the Early Prediction of Cost and Time Outcomes. In Proceedings of the Construction Research Congress 2014: Construction in a Global Network; American Society of Civil Engineers (ASCE): Reston, VA, USA, 2014; pp. 2306–2314. [ Google Scholar ]
- Tajziyehchi, N.; Moshirpour, M.; Jergeas, G.; Sadeghpour, F. A Predictive Model of Cost Growth in Construction Projects Using Feature Selection. In Proceedings of the 2020 IEEE Third International Conference on Artificial Intelligence and Knowledge Engineering (AIKE), Laguna Hills, CA, USA, 9–13 December 2020; IEEE: Piscataway, NJ, USA, 2020; pp. 142–147. [ Google Scholar ]
- Ajayi, A.A.; Mohamed, S.F. Cost Performance and Change Managementcapability Level of Contractors: A Relational Quantitative Study. Adv. Sci. Lett. 2015 , 21 , 1553–1556. [ Google Scholar ] [ CrossRef ]
- Sibiya, M.; Aigbavboa, C.; Thwala, W. Construction Projects’ Key Performance Indicators: A Case of the South African Construction Industry. In Proceedings of the ICCREM 2015, American Society of Civil Engineers, Luleå, Sweden, 28 September 2015; pp. 954–960. [ Google Scholar ]
- Nwadigo, O.; Naismith, N.N.; Ghaffarianhoseini, A.; Ghaffarian Hoseini, A.; Tookey, J. Dynamic Bayesian Network Modelling for Predicting Adaptability of Time Performance during Time Influencing Factors Disruptions in Construction Enterprise. ECAM 2021 , 28 , 2994–3013. [ Google Scholar ] [ CrossRef ]
- Musselwhite, D.; Gledson, B.; Greenwood, D. Matters affecting construction project-level planning effectiveness: A literature review. In Proceedings of the 37th Annual ARCOM Conference, ARCOM 2021, Virtual, Glasgow, UK, 6 September 2021; pp. 542–551. [ Google Scholar ]
- Perera, B.A.K.S.; Perera, C.S.R.; Jayalath, C. Contractor’s Perspective on Key Performance Indicators of Cost Control in Asian, Middle Eastern, and European Construction Projects. Int. J. Constr. Educ. Res. 2022 , 18 , 217–233. [ Google Scholar ] [ CrossRef ]
- Safapour, E.; Kermanshachi, S. Investigation and Analysis of Human, Organizational, and Project Based Rework Indicators in Construction Projects. In Proceedings of the Computing in Civil Engineering 2019, American Society of Civil Engineers, Atlanta, GA, USA, 13 June 2019; pp. 505–512. [ Google Scholar ]
- Bingol, B.N.; Polat, G. Measuring Managerial Capability of Subcontractors Using a KPI Model. Procedia Eng. 2017 , 196 , 68–75. [ Google Scholar ] [ CrossRef ]
- Chidiebere, E.E.; Ebhohimen, I.J. Impact of Rework on Building Project and Organisation Performance: A View of Construction Professionals in Nigeria. IJSCET 2018 , 9 , 29–43. [ Google Scholar ] [ CrossRef ]
- Nokulunga, M.; Didi, T.; Clinton, A. Cost of Poor Quality in Construction Projects in Swaziland. In Proceedings of the International Conference on Industrial Engineering and Operations Management, Washington, DC, USA, 27–29 September 2018. [ Google Scholar ]
- Koo, H.J.; O’Connor, J.; Sprouse, R. Analyzing the Characteristics of Design Defect Leading Indicators on Building Construction Projects. Int. J. Constr. Manag. 2024 , 24 , 834–842. [ Google Scholar ] [ CrossRef ]
- Heydari, M.; Heravi, G. A BIM-Based Framework for Optimization and Assessment of Buildings’ Cost and Carbon Emissions. J. Build. Eng. 2023 , 79 , 107762. [ Google Scholar ] [ CrossRef ]
- Zarei, B.; Sharifi, H.; Chaghouee, Y. Delay Causes Analysis in Complex Construction Projects: A Semantic Network Analysis Approach. Prod. Plan. Control 2018 , 29 , 29–40. [ Google Scholar ] [ CrossRef ]
- Mitchell, D. 5D BIM: Creating Cost Certainty and Better Buildings. In eWork and eBusiness in Architecture, Engineering and Construction—Proceedings of the European Conference on Product and Process Modelling 2012 ; Routledge: Oxfordshire, UK, 2012; pp. 253–258. [ Google Scholar ]
- Oyebisi, S.O.; Okeke, C.A.; Alayande, T.A.; Oluwafemi, J.O. Performance Evaluation in a Construction Project: An Empirical Study of Canaan City Housing Estate, Ota, Nigeria. Int. J. Qual. Eng. Technol. 2019 , 7 , 385–405. [ Google Scholar ] [ CrossRef ]
- Baldrich Aragó, A.; Roig Hernando, J.; Llovera Saez, F.J.; Coll Bertran, J. Quantity Surveying and BIM 5D. Its Implementation and Analysis Based on a Case Study Approach in Spain. J. Build. Eng. 2021 , 44 , 103234. [ Google Scholar ] [ CrossRef ]
Click here to enlarge figure
| No. | Questions |
|---|---|
| 1 | How has 5D BIM evolved in the construction industry over the past decade? |
| 2 | Which factors significantly influence the adoption of 5D BIM in the construction industry? |
| 3 | In what ways does 5D BIM impact project performance indicators? |
| Search string | (TITLE-ABS-KEY (“5D BIM”) OR TITLE-ABS-KEY (“BIM 5D”) OR TITLE-ABS-KEY (“5D Building Information Modeling”) OR TITLE-ABS-KEY (“the fifth dimension of BIM”) OR TITLE-ABS-KEY(“5 Dimensional Building Information Modeling”) OR TITLE-ABS-KEY(“Building Information Modeling 5D”) OR TITLE-ABS-KEY (“5D”) AND TITLE-ABS-KEY (“BIM”)) AND ((EXCLUDE (PUBYEAR, 2007) OR EXCLUDE (PUBYEAR, 2008) OR EXCLUDE (PUBYEAR, 2010) OR EXCLUDE (PUBYEAR, 2011) OR EXCLUDE (PUBYEAR, 2012) OR EXCLUDE (PUBYEAR, 2013) OR EXCLUDE (PUBYEAR, 2024)) |
| No. | Eligibility for Inclusion |
|---|---|
| 1 | Studies addressing the topic of 5D BIM or other synonyms |
| 2 | Studies published in the English language |
| 3 | Studies directly related to construction |
| 4 | Peer-reviewed publications (to ensure the inclusion of high-quality research) |
| 5 | Studies with a length of at least three pages |
| 6 | Articles with an explicit research title, abstract, and keywords |
| Software/Tool | Function (s) | Reference |
|---|---|---|
| VOSviewer 1.6.20 | Visualization and analysis of SLR data | [ ] |
| CiteSpace v.6.2.R6 (64-bit) Advanced | SLR cluster analysis/development path recording | [ ] |
| Microsoft Excel | Gathering, preserving, and displaying data | [ ] |
| Zotero 6.0.36 | Literature management | [ ] |
| Author | Documents | Total Citations | Proportion |
|---|---|---|---|
| Hosseini, M. Reza | 6 | 285 | 2.70% |
| Abrishami, Sepehr | 5 | 273 | 2.25% |
| Elghaish, Faris | 5 | 224 | 2.25% |
| Gaterell, Mark | 3 | 100 | 1.35% |
| Li, Hua | 3 | 27 | 1.35% |
| Brioso, Xavier | 3 | 13 | 1.35% |
| Pan, Yangshao | 3 | 9 | 1.35% |
| Guan, Changsheng | 3 | 2 | 1.35% |
| Vitasek, Stanislav | 3 | 6 | 1.35% |
| Factor Category | Sub-Categories | References |
|---|---|---|
| People/operational factors | Experts with training in operating tools | [ , , , , , , ] |
| Awareness of the project’s scope | [ , , , ] | |
| Prior experience partnering on 5D BIM projects | [ , ] | |
| Willingness to use 5D BIM | [ , , , , ] | |
| Collaboration concept among relevant stakeholders | [ , , , , , , ] | |
| Technological factors | Capacity of technology infrastructure | [ , , , ] |
| Conflicting implementation strategies of conventional approaches and 5D BIM | [ , , ] | |
| Availability of IT support | [ , , ] | |
| Compatibility with current industry standards | [ , ] | |
| Compatibility between software | [ , , , ] | |
| Organizational factors | Awareness of company | [ , , , , ] |
| Rationalization of the organizational structure of construction projects | [ , ] | |
| Constructability | [ , ] | |
| Level of project data management | [ , ] | |
| Costs related to BIM technology | [ , , , , , ] | |
| Project-related factors | Provision of 3D modeling/design | [ , , , ] |
| Provision of 4D modeling/schedule of constructionactivities | [ , ] | |
| Difficulty in checking documents caused by conflict detection | [ , , ] | |
| Incomplete/inaccurate data | [ , , , ] | |
| Predictability of project outcomes | [ , , ] | |
| Environmental factors | Market demand | [ , ] |
| Increasing competition in the construction industry | [ , ] | |
| Demand for sustainable urbanization | [ , , ] | |
| Business situation | [ , ] | |
| Cultural resistance preventing adoption | [ , , ] | |
| Strategy/government policy | Standards and guidelines related to BIM | [ , , , , , , , , ] |
| Contract standards for projects with BIM | [ , , , , ] | |
| Dispute settlement mechanisms for projects with BIM | [ , , ] | |
| Publicity and promotion for BIM | [ , , , ] | |
| Protection for intellectual property rights related to 5D BIM | [ , , ] |
| Factor Category | Sub-Category | References |
|---|---|---|
| Project cost performance | Cost estimation | [ , , , , , , , , , , , , , , , , , ] |
| Cost control | [ , , , , , , , , , , , , , , ] | |
| Cost budgeting | [ , , , , , , , , , , ] | |
| Quantity takeoff | [ , , , , , , , ] | |
| Claims | [ , , , ] | |
| Project time performance | Enhanced decision making | [ , , , ] |
| Scheduled variance analysis | [ , , , , , , ] | |
| Shorter project times through coordination | [ , , ] | |
| Time risk management | [ , , , , , , ] | |
| Time-efficient construction delivery | [ , ] | |
| Project quality performance | Sustainable development of the construction project | [ , ] |
| Continuous improvement/process optimization | [ , , , ] | |
| Quality of data documentation | [ , , , ] | |
| Reductions in defects and quality errors | [ , ] | |
| Satisfactory workplace environment | [ , , ] |
| Category | KPIs | References |
|---|---|---|
| Project cost performance indicators | Cost performance | [ , ] |
| Cost predictability | [ , ] | |
| Project cost growth | [ ] | |
| Change cost factor | [ , ] | |
| Project budget factor | [ , ] | |
| Project time performance indicators | Time predictability | [ , ] |
| Schedule performance | [ , ] | |
| Change in project schedule | [ , ] | |
| Project quality performance indicators | Quality/high-quality performance | [ , ] |
| Rework | [ , ] | |
| Defects and quality errors | [ , ] |
| The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
Share and Cite
Sun, H.; Khoo, T.J.; Esa, M.; Mahdiyar, A.; Li, J. Critical Factors Driving Construction Project Performance in Integrated 5D Building Information Modeling. Buildings 2024 , 14 , 2807. https://doi.org/10.3390/buildings14092807
Sun H, Khoo TJ, Esa M, Mahdiyar A, Li J. Critical Factors Driving Construction Project Performance in Integrated 5D Building Information Modeling. Buildings . 2024; 14(9):2807. https://doi.org/10.3390/buildings14092807
Sun, Hui, Terh Jing Khoo, Muneera Esa, Amir Mahdiyar, and Jiguang Li. 2024. "Critical Factors Driving Construction Project Performance in Integrated 5D Building Information Modeling" Buildings 14, no. 9: 2807. https://doi.org/10.3390/buildings14092807
Article Metrics
Article access statistics, further information, mdpi initiatives, follow mdpi.

Subscribe to receive issue release notifications and newsletters from MDPI journals
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- SAGE Open Med
End-of-life decision making by family caregivers of persons with advanced dementia: A literature review of decision aids
1 School of Nursing, The University of Texas at Austin, Austin, TX, USA
2 School of Information, The University of Texas at Austin, Austin, TX, USA
Amy S Berkley
Kenneth r fleischmann, jane dimmitt champion, kolina s koltai, objectives:.
To investigate existing knowledge in the literature about end-of-life decision making by family caregivers of persons with dementia, focusing on decision aids for caregivers of persons with advanced dementia, and to identify gaps in the literature that can guide future research.
A literature review through systematic searches in PubMed, CINAHL Plus with Full Text, and PsycINFO was conducted in February 2018; publications with full text in English and published in the past 10 years were selected in multiple steps.
The final sample included five decision aids with predominantly Caucasian participants; three of them had control groups, and three used audiovisual technology in presenting the intervention materials. No other technology was used in any intervention. Existing interventions lacked tailoring of information to caregivers’ preferences for different types and amounts of information necessary to make decisions consistent with patients’ values.
Conclusion:
Research is needed in exploring the use of technology in decision aids that could provide tailored information to facilitate caregivers’ decision making. More diverse samples are needed.
Introduction
Dementia, a collection of symptoms involving cognitive impairment severe enough to interfere with daily living, is a major public health issue worldwide. In the United States, as the number and proportion of older adults increase, the number of persons living with dementia is projected to increase from 5.4 million in 2016 to 13.8 million in 2050, with the greatest growth among those in advanced stage. 1 Dementia is the sixth leading cause of death in the United States, the only one for which there is currently no treatment or cure. 1
The very nature of dementia requires heavy family involvement, which can be stressful and challenging. 2 Negative consequences for caregivers’ own health and well-being have been well documented. 3 , 4 Even after placing a person with dementia in a care facility (e.g. nursing homes), the caregiver continues to be involved, supervising the care of the person with dementia at the facility and serving as a surrogate decision maker for important legal, financial, and healthcare decisions. 5 The ethical and emotional responsibilities of caregivers are great, especially when decisions involve end-of-life (EOL) care. Such decisions are often high-stake, value-laden, and complex (e.g. withdrawing artificial nutrition/hydration or antibiotics, or enrolling in hospice). 6 – 9
In response to the growing number of persons with dementia at EOL and the complexity of decision-making responsibilities of caregivers, researchers have begun to develop and test interventions that can support family caregivers’ decision making for persons with dementia. This is done primarily through the use of decision aids, which are structured tools designed to help patients and their families make specific, informed healthcare choices consistent with patients’ values. 10 Decision aids typically contain pertinent educational information about a patient’s condition or disease and about the advantages and disadvantages of treatment options; assessment tools that help individuals clarify their values by asking users to rate their perceptions of the benefits and disadvantages of different treatment or options; and advice about the decision-making process. 9 Formats of decision aids vary from video to print information to help decision makers consider how risk, uncertainty, and values may affect clinical and care choices. 7 Developed as an adjunct to, not in lieu of, consultation and counseling from healthcare providers, decision aids have been shown to improve knowledge, engagement in shared decision making, satisfaction with decisions, and compliance with treatment plans. 11 Decision aids are typically designed to be brief and simple to administer, thus addressing a major barrier to providers’ engagement with patients and their families in shared decision making—that is, time constraints.
To our knowledge, no literature review exists to systematically compare and summarize the characteristics of existing decision aids for caregivers of persons with advanced dementia. To address this gap in the literature, this literature review aims to systematically investigate existing knowledge in the literature about EOL decision making in family caregivers of persons with dementia, focusing on the types of decision aids available for family caregivers of persons with advanced dementia, and to identify gaps in the literature that can guide future research (e.g. we intend to develop and test a mobile technology–based decision aid for caregivers). Our overarching research question (RQ) was. “What were the main characteristics of existing decision aids for family caregivers of persons with advanced dementia?” Under this overarching RQ, we had two specific RQs: How might existing decision aids have involved tailoring, specifically based on patients’ values and their caregivers’ information preferences? (RQa) And what types of technology were used in existing decision aids for family caregivers of persons with advanced dementia to obtain desired information (RQb)?
Article selection
Following strategies used in earlier literature review studies, 12 – 14 we performed three rounds of systematic selection in the PubMed database: (a) keyword search in Titles/Abstracts and MeSH terms, (b) screening of titles and abstracts, and (c) screening of full-text articles. The searches were performed on 5 February 2018.
Round 1: keyword search
We used the following four sets of keywords to search the Title/Abstract field in PubMed: (“dementia” OR “Alzheimer’s”) AND (“decision aid*”) AND (“caregiver*” OR “proxy” OR “proxies” OR “surrogate*”) AND (“end of life” OR “advanced dementia”). We used the following PubMed filters: (1) Text availability: Full text, (2) Publication dates: 10 years, and (3) Species: Humans. This process produced a total of seven articles. Next, we searched in MeSH terms in PubMed, using the same search terms mentioned above (except for “surrogate*,” which was not a MeSH term) and with the same three built-in filters (full text available, published in the past 10 years, and humans). A total of three results were found. Combining the results from these two searches yielded a total of 10 non-duplicate results.
In addition to PubMed, we also searched in two other databases on the same day: CINAHL Plus with Full Text and PsycINFO. We used the same four sets of keywords that we used for PubMed to search in the Abstract field of these two additional databases. We also added the following built-in filters (“Limiters”) when searching in these two additional databases: Full Text, References Available, English Language, Peer Reviewed, Research Article, Human, and Journal Article. Our searches found no additional results than we had already found in PubMed. Thus, the 10 results found during this round were used in the subsequent rounds of screening.
Round 2: screening the titles and abstracts
Author B.X. manually screened the titles and abstracts of the 10 articles to ensure they covered all four key aspects that guided our first round of searches, with the following further criteria predetermined by authors B.X., J.K., and K.R.F.:
- Articles reporting original data were included; those that did not report original data were excluded.
- Articles in which family caregivers of dementia patients constituted at least part of the study sample were included. For example, a study that included both healthcare providers and family caregivers was included, whereas a study that had only healthcare providers in the study sample without any family caregivers was excluded.
After these criteria were applied, five articles remained. The full text of the remaining five articles was examined and confirmed that they all met the criteria. This final sample comprised empirical studies published over the last 10 years that focused on decision aids for the EOL decision making of family caregivers of persons with advanced dementia. The selection process is illustrated in Figure 1 .

Systematic selection process.
Round 3: coding the full-text articles
We reviewed the full text of the final five articles and coded them by two independent coders (B.X. and A.S.B.) using a coding sheet predetermined by authors B.X., J.K., and K.R.F. based on prior work. 12 – 14 Discrepancies between the two independent coders’ coding were minimal. After all five articles were coded, key information from each article was summarized.
Key characteristics of the studies in our final sample are summarized in Table 1 .
Summary of the studies in the final sample.
| Reference | Purpose/aims | Sample | Research design | Intervention; technology used | Key findings |
|---|---|---|---|---|---|
| Einterz et al. | To explore the feasibility of a decision aid intervention for caregivers and to generate preliminary evidence on the intervention’s effect on quality of communication and decision making | = 18 pairs of persons with (moderate to severe) dementia and their caregivers | Pre-/posttest; no control | Each caregiver viewed the decision aid video and participated in a structured care plan meeting with an interdisciplinary team Technology used: video | The intervention was feasible and relevant for the target population; it improved caregivers’ knowledge, quality of communication, and particularly quality of communication at EOL with providers |
| Hanson et al. | To describe the protocols of a decision aid intervention, challenges, and strategies for recruitment and retention, and approaches to ensuring research ethics | = 256 dyads of persons with advanced dementia and their caregivers | Cluster randomized controlled trial | Caregivers reviewed information (in print or audio) about dementia, feeding options in advanced dementia. Controls received usual care Technology used: audio narration with words on a computer screen for participants with impaired vision or limited literacy | The research team used multiple strategies to recruit and retain sites and participants; successfully enrolled 256 dyads, and 99% of them completed the 3-month study period; recognizing persons with advanced dementia as a vulnerable population is important for ensuring research ethics |
| Hanson et al. | To test the efficacy of a decision aid (the same one introduced in Hanson et al. 2010) in improving the quality of decision making about feeding options in advanced dementia | = 256 dyads of persons with advanced dementia and their caregivers | Cluster randomized controlled trial | Same as reported in Hanson et al. | Improved knowledge among caregivers in the intervention group (16.8 vs 15.1, < .001); after 3 months, caregivers in the intervention group had lower Decisional Conflict Scale scores than did those in the control group (1.65 vs 1.90, < .001) and had more discussions about feeding options with a healthcare provider (46% vs 33%, = .04). Persons with advanced dementia in the intervention group were more likely to receive a dysphagia diet (89% vs 76%, = .04). No statistically significant difference in tube feeding between the intervention and control groups |
| Hanson et al. | To describe strategies used to monitor and promote the fidelity of a decision aid intervention | = 151 dyads of persons with dementia and their caregivers | Cluster randomized controlled trial | Intervention group received (1) a 20-min video decision aid about care choices in advanced dementia, and (2) a structured nursing home care plan meeting to address goals of care. Three goals were covered: prolonging life, supporting function, and improving comfort; Control: viewed a 20-min informational video about dementia and participated in usual care plan meetings with staff Technology used: video | Intervention fidelity strategies used in the study enabled providers to implement a decision aid intervention for caregivers of persons with advanced dementia |
| Snyder et al. | Aim 1: To describe caregivers’ perceptions of feeding options for their relatives with advanced dementia living in skilled nursing facilities; Aim 2: To explore how a decision aid might change caregivers’ knowledge about feeding options, expectations of tube feeding benefits, decisional conflict, and preferred feeding method | Aim 1: = 255; Aim 2: = 126 (only caregivers in the intervention group were studied for Aim 2) | Aim 1: semi-structured interviews with open-ended questions; Aim 2: pre-/posttest; no control | A decision aid (in audio, visual, and print formats) provided caregivers in the intervention group with information about dementia, feeding problems, pros and cons of feeding options, and surrogates’ role in decision-making Audio-visual technology | From pre- to posttest, participants in the intervention group showed improved knowledge (15.5 vs 16.8; < .001), decreased expectation of tube feeding benefits (2.73 vs 2.32; = .001), and decreased decisional conflict (2.24 vs 1.91; < .001). Their preference for assisted oral feeding did not change, but certainty about their choice improved (1.35 vs 1.05; = .016), suggesting the efficacy of using this decision aid in improving caregiver decision making about feeding options in the care for advanced dementia patients |
EOL: end-of-life.
The studies’ samples varied widely, from 36 to 512. All interventions consisted of predominately Caucasian participants. Two of the interventions in our final sample did not have control groups; 7 , 17 the others did. 2 , 15 , 16 All interventions used audiovisual technology—intervention materials delivered in an audio/video format in addition to a conventional print format. No other technology was used in any of the interventions. The interventions focused on factors affecting typical EOL decision-making processes and outcomes (especially feeding-related), that is, knowledge about feeding options, communication skills, decisional conflicts, and decisions about treatment.
The interventions all involved delivering educational materials to family caregivers, although differences existed in how those materials were delivered. Some of the interventions featured providing participants with the same generic materials to review on their own with no other intervention elements. 2 , 15 , 17 Other interventions 7 , 16 involved participants reviewing the materials individually, followed by a structured care plan meeting with the staff, with the latter presumably enabling opportunities to provide information tailored to caregivers’ unique circumstances. Notably though, none of the interventions specifically acknowledged the need for tailoring of information to the needs of caregivers or patients in different situations.
The studies in this review involved several different instruments to measure a range of concepts, most commonly knowledge, communication skills, decisional conflict, comfort with knowledge, confidence with treatment, and satisfaction with care. Many of these instruments were developed specifically for these interventions and require further psychometric testing in different populations and contexts. The studies that focused on the efficacy of their interventions 2 , 17 reported their interventions were effective in at least one of their outcome measures, that is, improved knowledge, quality of communication, or decreased decisional conflict. One intervention found statistically significant difference in tube feeding between the intervention and control groups. 2
It should be noted that the reviewed articles provided insufficient information about the interventions, making it difficult to extract interventions’ details and make comparisons across the studies. The description of an intervention could be as simple as one sentence, for example: 17 “Intervention: For intervention surrogates only, an audiovisual-print decision aid provided information on dementia, feeding problems in dementia, advantages and disadvantages of feeding tubes or assisted oral feeding options, and the role of surrogates in making these decisions” (p. 114).
The small number of interventions in our final sample suggests that decision aids supporting EOL decision making by caregivers of persons with dementia is currently understudied and in need of extensive research. Our ability to make comparisons between studies was limited because of the differing methods employed and different types of decision aids examined. An added challenge is that insufficient information was provided in the publications with regard to various specifics about the decision aids. Future studies should report details of the interventions such as the amount of information provided (e.g., number of pages of print materials, minutes of audio/video provided, approximate time required to review the information) and the intervention’s delivery mode (e.g. group size or individual information review, presence or absence of tailored in-person guidance).
Feeding tube-related issues faced by caregivers of people with advanced dementia were the primary focus of decision aids identified in this review of empirical studies. 5 , 7 As the condition of the person with dementia deteriorates, decisions involve more emotionally laden issues such as advance care planning or resuscitation orders. 6 , 18 Such decisions are difficult, because they require the caregiver to exercise substituted judgment or follow best interest standards to make decisions on behalf of the person with dementia; yet, it is often difficult to know what the wishes of a person with dementia are or what the best course of treatments are, given the uncertain, protracted nature of dementia’s progression. 6 , 18 , 19
A lack of focus on tailoring to caregivers’ information preferences necessary to ensure decision making consistent with patients’ values
The interventional studies in our sample generally concluded that their decision aids helped caregivers with their decision making. However, a major limitation of existing decision aids is that they typically do not take into account caregivers’ preferences for different types and amounts of information that are necessary to ensure decision making consistent with patients’ values. The decision aids in these studies were typically limited by a one-directional approach: the types and amounts of information provided were predetermined by researchers/providers, instead of tailored to the information preferences of individual caregivers. Subsequently, caregivers might not receive the right types and/or amounts of information necessary for making decisions consistent with patients’ values. This lack of focus on values and preferences may reflect an assumption in the current medical model that patients have well-formed EOL care preferences consistent with their values and that caregivers can ascertain what those preferences are, or that caregivers are fully informed of how different care decisions may, or may not, support their loved ones’ values and EOL preferences. However, such an assumption frequently fails to reflect reality. Patients and their spouses can differ significantly in EOL care preferences, with patients more likely to prefer additional treatment than their spouses. 20 This is consistent with findings from earlier research showing low to modest congruence between patients and caregivers. 21 When potential caregivers were asked in scenarios to predict patients’ preferences, accuracy was highest when patients’ current health status was considered and lowest in scenarios of stroke and dementia; caregivers reported that EOL discussions failed to occur for a number of reasons, but mostly because of family belief structures and personalities. 22
In a study that compared hypothetical EOL decision-making vignettes between relatives and professionals acting as patient surrogates, researchers found that situational variables such as the patient’s current behavior and the views of healthcare professionals and family members had higher impacts on decisions than did the patient’s prior statements or life attitudes. 8 Other researchers who have looked at EOL decision-making scenarios with healthy older adults have found other factors that influence caregivers’ decision making, such as caregivers’ own current state of health, health literacy, and communication with healthcare providers. 8 , 22 – 24 Even when patients’ care preferences are known to caregivers, they might still not be implemented, due to factors such as attempting to achieve family consensus. 25 Evidence from 228 community-dwelling family care dyads showed that 25% of cargivers underestimated the importance of everyday care values of persons with dementia, suggesting incongruence between patients’ and caregivers’ values. 26 If and how these factors affect caregiver decision making deserves further examination and implementation in decision aids.
Underutilization of technology
Despite advances in information and communication technology, none of the decision aids in our sample involved any kind of technology beyond audio/video as a part of an intervention. Existing decision aids are typically delivered to caregivers in print (typically a hardcopy workbook), sometimes coupled with in-person discussions with trained providers in a group or individually. The printed materials’ content varied; topics included information about advanced dementia, the role of the surrogate decision maker, EOL care options and their pros and cons, common community services available, and pointers toward further information. Presented in a static print or audio/video format, such rich information can be overwhelming, and some of it may even be irrelevant to individuals with unique values, preferences, or other circumstances. Tailoring interventions to meet the needs of particular groups is critical, and such interactive information can best be delivered using more advanced technology such as a website or mobile app that would allow for interactive use beyond the scope of a particular class discussion setting. Doing so is important because caregivers are likely to think of additional questions while engaging in the everyday process of caring for persons with dementia. Unfortunately, none of the studies reviewed employed such advanced technology in their interventions.
For technology to be useful in decision aids for caregivers of persons with dementia, an important point to note is that caregivers (e.g. spouses of persons with dementia) may be of older ages themselves and/or facing other challenges (e.g. limited literacy). As such, it is critical that any technology used in a decision aid is easy to use for caregivers with limited technology experience and skills. Age-appropriate training and technical support will be necessary as well. Furthermore, integrating technology in decision aids is not to completely replace human interactions, as interactions with healthcare professionals can be instrumental to the caregiver’s decision-making process and outcomes.
The importance of advance care planning
A major topic commonly addressed in the literature was advance care planning for persons with dementia. Caregivers in various sample populations had taken on this responsibility with little or no knowledge of the values and desires of their relatives regarding EOL care and management. Decision aids have been designed to encourage caregivers to prepare advance directives and/or discuss possible treatment limitations with healthcare providers. Advance care plans are themselves a type of decision aid because they educate and prepare caregivers for the progression that they can expect as their relatives decline toward the end of life. 6 , 7 In an ideal situation, the demands of an advance directive or any kind of advance care planning would be discussed with the family member while the person is still able to express his or her wishes about EOL care. Decisional conflict was greater with caregivers who were struggling to make sense of their relatives’ wishes, and the literature showed that decision aids did help in these situations. 5 – 7
An important aspect of advance care planning that requires greater acceptance is the use of do not hospitalize (DNH) orders: 27 barriers to these orders included a “perceived lack of physician involvement in decision-making and limited understanding of DNH orders and the resident’s prognosis” (p. 1568). Meanwhile, evidence also exists that advance directives (specifically, euthanasia directives in the Netherlands) were hardly ever actually adhered to by physicians and family members after they had been completed, 18 although such a phenomenon may have limited generalizability given its specific context.
The controversies around tube feeding and use of antibiotics
Feeding is one of the most contentious areas of decision making for persons with dementia. Decision aids typically included content designed to help caregivers understand the issues surrounding feeding, percutaneous endoscopic gastronomy tube insertion, and alternative treatments. Controlled observational studies show that tube feeding does not improve survival, aspiration, or wound healing; however, this information is not routinely shared with decision makers, and families and professionals alike may have unrealistically high expectations for benefits from tube feeding. 2 The decision aids in this literature review frequently involved improving caregiver knowledge about problems with tube feeding, separating the desire to support and “nourish” the person with dementia from the physical need to feed someone mechanically who has a limited life expectancy. 2 , 5 , 7 However, the decision aids did not assess caregivers’ preferences for different types and amounts of information beforehand and presented little opportunity to include them in the decision-making process.
Related to tube feeding is the question of treatment for infections. Treatment with antibiotics frequently brings its own set of complications, including gastrointestinal upset and other side effects. Also, transferring a person with dementia can worsen disorientation because it places the person in an unfamiliar environment. In a study that examined caregivers’ involvement in decisions about whether or not to treat minor infections in persons with dementia and how aggressively to do so, the researchers found that caregivers participated only about half of the time. 5 Studies of family members’ perceptions of the EOL decision-making process and outcomes suggest important opportunities for the improvement of decision making in EOL care. 28
The need for research on diverse populations
Using racially/ethnically homogeneous samples—that consisted of predominately Caucasian participants—was a major limitation in studies in this review. Decision making is culturally sensitive, and more diverse samples would enhance our knowledge in this area. In addition, ethnically and racially diverse samples may well point out a need for more flexible, customizable decision aids. For instance, prior evidence suggests that African Americans were less likely than Caucasian Americans to have advance care plans, and implementing programs in the community was effective in enhancing success because participants felt comfortable discussing sensitive topics in environments they could trust. 6 A study of Hong Kong Chinese family caregivers found that reliance on collective family decisions significantly affected choices of caregivers and that filial piety and possible confrontation from relatives, combined with a lack of knowledge about life-sustaining treatments, worsened decision-making stresses. 29 Compared with Caucasian elders and their caregivers, non-Caucasians and individuals with greater financial difficulty had stronger preferences for life-prolonging treatment. 23
People from ethnic minority groups may prefer different EOL treatments and are, due to disparities and differences in values and beliefs, less likely to have advance directives (e.g. members of specific ethnic groups may have strong cultural taboos against open and direct discussion about death and dying 30 , 31 ). The experiences of ethnic minority caregivers differ as well (e.g. higher levels of depression and stress among Hispanic caregivers than among non-Hispanic Caucasians 32 ). Caregivers and patients in rural areas also face unique challenges: they tend to be older, in worse health, with greater financial burden and limited access to providers and support services, experiencing stigma of dementia and a lack of privacy. 33 – 38 The coping styles of caregivers of persons with dementia in rural areas are often different from those in urban areas, suggesting unique needs. 38 The significant health disparities and characteristics among caregivers who are members of racial/ethnic minority groups and/or live in rural areas call for effective interventions tailored to their unique needs and circumstances.
Limitations of this literature review
This literature review has limitations. We searched in only three databases with specific inclusion/exclusion criteria. While these criteria were necessary for our specific purposes (i.e. preparing for a larger intervention study involving using interactive technology to provide tailored decision aids for caregivers of persons with advanced dementia), using these criteria led to only five relevant articles in our final sample. One possible reason is that we used search terms common to the US context, which might not necessarily be common terms used in other national contexts. Subsequently, our study is geared more toward the US context. Also, our searches focused on decision aids, for the purpose of comparing and analyzing what has been done and what has not with existing decision aid interventions. It was beyond the scope of this current literature review to examine the literature on broader issues related to decision making.
This literature review has identified several issues that call for attention. Few decision aids exist for caregivers’ EOL decision making for persons with dementia. Existing studies lacked a focus on caregivers’ preferences for different types and amounts of information necessary to ensure decision making consistent with patients’ values. Advance care planning for persons with dementia is a major challenge for caregivers. Ideally, the demands of an advance directive or any kind of advance care planning would have been discussed with the person with dementia while he or she was still able to express such wishes. Yet, this is often not the case. Developers of decision aids should strive to find ways to incorporate values and preferences in decision making. None of the studies mentioned any tailoring or acknowledged the need for specific tailoring of information to the individual situations of patients or caregivers. Important variables for tailoring such as values and information preferences were typically not assessed before or during the implementation of decision aids. While recent technological developments enable great tailoring of information, existing decision aids have largely underutilized such tailoring potential. Future research should investigate whether/how we could take full advantage of recent technological developments in the design and implementation of future decision aids (that are easy to use by caregivers with limited technology literacy). Finally, existing studies involved samples predominantly of non-Hispanic Caucasians. Since EOL decision making is culturally sensitive, future research should place special emphasis on ethnically and racially diverse samples and develop tailored and interactive decision aids for underrepresented groups.
Acknowledgments
This work was part of a Pilot Research Grant from the Population Health Initiative at the University of Texas at Austin, entitled Values and Preferences in Dementia Family Caregivers’ End-of-Life Decision-Making ; Principal Investigator (PI): Bo Xie; Co-PIs: Ken Fleischmann and Jung Kwak.
Declaration of conflicting interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was part of a Pilot Research Grant from the Population Health Initiative at the University of Texas at Austin, entitled Values and Preferences in Dementia Family Caregivers’ End-of-Life Decision-Making ; Principal Investigator (PI): Bo Xie; Co-PIs: Ken Fleischmann and Jung Kwak.


IMAGES
VIDEO
COMMENTS
Whereas decision aids such as decision support systems (DSS) can be beneficial in stressful scenarios, decision makers sometimes misuse them during decision making, leading to suboptimal outcomes. ... Stressed decision makers and use of decision aids: A literature review and conceptual model. Information Technology & People, 33(2), 710-754 ...
Previous versions of the International Patient Decision Aid Standards (IPDAS) began to map this diversity, with the 2013 update offering a model process based on the authors' review of the literature. Since that last update, there has been increasing recognition of the importance of engaging the users of decision aids in their development.
Limitations of traditional decision aids. For the past two decades enthusiasts have advocated decision aids to facilitate shared decision making, and over 500 have been developed.15 16 A systematic review of 115 randomised trials showed that their use was associated with a 13% absolute increase in patients' knowledge scores and an 82% relative increase in accurate expectations of possible ...
The Cochrane Review of Patient Decision Aids: This Cochrane review, initiated in 2003, plays a pivotal role in summarizing the existing evidence on the impact of these tools. It is the most referenced review in the Cochrane collection (over 8400 citations). In their 2024 update, Dr. Logan Trenaman and the Cochrane team identified 104 new ...
There has been a steady increase in the development and evaluation of PDAs over the last 15 years (O'Connor et al., 1999; Stacey et al., 2014).Patients given decision aids in the studies took a more active role in making the decision and had improved knowledge, more realistic expectations of the benefits and side-effects of their options, lower decisional conflict, and were more likely to ...
Literature search. We reviewed all studies included in the Cochrane review entitled "Decision aids for people facing health treatment or screening decisions" published in April 2017 (literature search conducted on 24 April 2015). 4 The Cochrane review captured trials of decision aids for people facing health treatment or screening decisions. The majority of studies in the Cochrane review ...
To review the literature and provide a summary of best practices for the design, implementation, and assessment of decision aids. Methods. ... Interestingly, in their review, decision aids had a variable effect on consultation length, from shorter to longer durations, so perhaps confronting the perception of time consumption would be important ...
Shared decision making (SDM) and patient-centered care require patients to actively participate in the decision-making process. Yet with the increasing number and complexity of cancer treatment options, it can be a challenge for patients to evaluate clinical information and make risk-benefit trade-offs to choose the most appropriate treatment. Clinicians face time constraints and ...
To provide further details about design and development methods (objective 1), we summarized findings from the extensive systematic review of literature published up to June 2017 by Vaisson et al. 3 To describe the application of user-centered design during the development of decision aids (objective 2), we conducted a focused review of patient ...
The purpose of this paper is to investigate the relationship between stress, decision making and decision aid use.,The authors conduct an extensive multi-disciplinary review of decision making and DSS use through the lens of stress and examine how stress, as perceived by decision makers, impacts their use or misuse of DSS even when such aids ...
Physician-Patient Relations*. Shared decision making (SDM) is a process within a patient centred consultation that involves both the patient and doctor discussing management options and agreeing on management decisions in partnership. Decision aids are designed to help patients understand the options relating to management for c ….
Decision aids for patients have recently been introduced in health care. A literature review was conducted to address the following research questions: 1) which types of decision aids have been developed?; 2) to what extent are they feasible, and acceptable to patients and health care providers?; 3) …
Decision aids are one type of tool that can be used to support the process of shared decision making between patients and their health professionals, a key element of person-centred care and health system improvements. A major update to the landmark Cochrane review on decision aids has just been published with an analysis of 209 studies ...
- Patient decision aids are pamphlets or videos used in person or online. They clearly identify the healthcare decision to be made, provide information on options (benefits and harms), and help people clarify what is most important to them. Decision aids are designed to enhance and supplement consultation with the clinician, not replace it.
Decision-aids are reported to increase knowledge, reduce decisional conflict, cause greater satisfaction with decision-making, support more realistic expectations, achieve a greater likelihood of ...
The evolution and rationale behind SDM and the evidence relating to outcomes, the types of decision aids available, and research relating to their use are discussed. Shared decision making (SDM) is a process within a patient centred consultation that involves both the patient and doctor discussing management options and agreeing on management decisions in partnership.
Request PDF | Stressed decision makers and use of decision aids: a literature review and conceptual model | Purpose Decision making is inherently stressful since the decision maker must choose ...
Nogués X, Carbonell MC, Canals L, Lizán L, Palacios S. Current situation of shared decision making in osteoporosis: A comprehensive literature review of patient decision aids and decision drivers. Health Sci Rep 2022; 5 :e849. 10.1002/hsr2.849 [ CrossRef ] [ Google Scholar ]
The aim of this systematic review is to investigate the causes and effects of decision fatigue from the existing literature that can be generalized across different organizational domains. A comprehensive literature search in three databases identified 589 articles on decision fatigue.
To address this gap in the literature, this literature review aims to systematically investigate existing knowledge in the literature about EOL decision making in family caregivers of persons with dementia, focusing on the types of decision aids available for family caregivers of persons with advanced dementia, and to identify gaps in the ...
For other testing and screening choices, mostly there were no differences between decision aids and usual care. The median effect of decision aids on length of consultation was 2.6 minutes longer (24 versus 21; 7.5% increase). The costs of the decision aid group were lower in two studies and similar to usual care in four studies.
Patient decision aids in this review improved patient knowledge, but decisional conflict scores were unchanged, possibly due to a ceiling effect. ... Literature 40: A decision aid for treatment options for severe aortic stenosis for patients deciding between TAVI and surgery 41: TAVI. SAVR. American College of Cardiology:
The median numbers of primary studies and participants assessed in a review were 10 (range, 2-35) and 4328 (range, 518-12,781), respectively (Table 1 and Appendix Table 1).RCTs dominated most of the included studies: 16 of 27 (59%) reviews included RCTs only (5/8 [63%], 5/8 [63%], and 4/7 [57%] reviews focused on prostate, breast, and colorectal cancer screening decision aids, respectively ...
Objectives: To investigate existing knowledge in the literature about end-of-life decision making by family caregivers of persons with dementia, focusing on decision aids for caregivers of persons with advanced dementia, and to identify gaps in the literature that can guide future research. Methods: A literature review through systematic searches in PubMed, CINAHL Plus with Full Text, and ...
This review summarizes the main scores, including MADIT trial-based Risk Stratification Score (MRSS) and Seattle Heart Failure Model (SHFM), which are based on randomized trials with a control group (HF medication only) and validated on large cohorts of 'real-world' HF patients. ... but could aid decision regarding prophylactic ICD in ...
This study presents a detailed case analysis of a 40-year-old male patient with hemophilia A and severe chronic elbow arthropathy, exploring the surgical challenges and outcomes within the context of the current literature. The patient, with a history of multiple comorbidities including Hodgkin's lymphoma and cardiomyopathy, exhibited significant joint damage and functional impairment. A ...
To our knowledge, no literature review exists to system-atically compare and summarize the characteristics of exist-ing decision aids for caregivers of persons with advanced dementia. To address this gap in the literature, this literature review aims to systematically investigate existing knowl-
Purchasing and procurement managers should make informed decisions in selecting materials at the right time, in sufficient quantities, and at affordable prices. Supplier selection and order allocation (SSOA) is a vital aspect of purchasing and procurement processes. In this research, the techniques and decision-making methods used in SSOA from peer-reviewed journals published from 2021 to 2023 ...
A thorough systematic literature review and qualitative analysis were conducted to achieve this goal. Relevant articles from the past decade (2014-2023) were examined from the Scopus and Web of Science databases, of which 222 were selected and screened using PRISMA procedures. ... and can aid them in making informed decisions .
This literature review has identified several issues that call for attention. Few decision aids exist for caregivers' EOL decision making for persons with dementia. Existing studies lacked a focus on caregivers' preferences for different types and amounts of information necessary to ensure decision making consistent with patients' values.